Pentane - Thermophysical Properties
Chemical, physical and thermal properties of pentane, also called n-pentane. Phase diagram included.
Pentane, C5 H12 , is a clear colorless liquid with a petroleum-like odor. It belongs to the organic class alkanes, and is naturally present in crude oils and condensates. It is a component of some fuels and is employed as a specialty solvent in the laboratory.
The boling point 36°C/97°F, and the vapors are heavier than air. Both the liquid an the vapor are flammable.
The phase diagram of pentane is shown below the table.
Chemical, physical and thermal properties of pentane :
Values are given for liquid at 25 oC (77 oF, 298 K) and 1 bara, if not other phase, temperature or pressure given.
For full table with Imperial Units - rotate the screen!
| Property | Value | Unit | Value | Unit | Value | Unit | Value | Unit | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Autoignition temperature | 533 | K | 260 | °C | 500 | °F | |||||
| Boiling Point | 309.2 | K | 36.06 | °C | 96.9 | °F | |||||
| Critical density | 3.22 | mol/dm3 | 232 | kg/m3 | 0.450 | slug/ft3 | 14.5 | lb/ft3 | |||
| Critical pressure | 3.36 | MPa=MN/m2 | 33.6 | bar | 33.2 | atm | 487 | psi=lbf/in2 | |||
| Critical temperature | 469.8 | K | 196.7 | °C | 386.0 | °F | |||||
| Critical volume | 311 | cm3/mol | 0.00431 | m3/kg | 2.22 | ft3/slug | 0.0690 | ft3/lb | |||
| Density | 8606 | mol/m3 | 620.9 | kg/m3 | 1.205 | slug/ft3 | 38.76 | lb/ft3 | |||
| Flammable, gas and liquid | yes | ||||||||||
| Flash point | 224 | K | -49 | °C | -56 | °F | |||||
| Gas constant, individual, R | 115.2 | J/kg K | 0.03201 | Wh/(kg K) | 689.1 | (ft lbf/slug °R) | 21.42 | (ft lbf/lb °R) | |||
| Gibbs free energy of formation (gas) | -8 | kJ/mol | -111 | kJ/kg | -48 | Btu/lb | |||||
| Heat (enthalpy) of combustion (gas) | -3535 | kJ/mol | -48996 | kJ/kg | -21.1 | Btu/lb | |||||
| Heat (enthalpy) of combustion (liquid) | -3509 | kJ/mol | -48636 | kJ/kg | -20.9 | Btu/lb | |||||
| Heat (enthalpy) of formation (gas) | -147.0 | kJ/mol | -2037 | kJ/kg | -876 | Btu/lb | |||||
| Heat (enthalpy) of formation (liquid) | -173 | kJ/mol | -2398 | kJ/kg | -1031 | Btu/lb | |||||
| Heat (enthalpy) of fusion at -202 °F/-130°C | 8.4 | kJ/mol | 116 | kJ/kg | 50.05 | Btu/lb | |||||
| Heat (enthalpy) of sublimation, at -202°F/-130°C | 42 | kJ/mol | 582 | kJ/kg | 250 | Btu/lb | |||||
| Heat (enthalpy) of evaporation | 26.4 | kJ/mol | 366 | kJ/kg | 157 | Btu/lb | |||||
| Heat capacity, Cp (gas) | 120.0 | J/mol K | 1.66 | kJ/kg K | 0.397 | Btu/lb°F or cal/g K | |||||
| Specific heat, Cp (liquid) | 168.0 | J/mol K | 2.33 | kJ/kg K | 0.556 | Btu/lb°F or cal/g K | |||||
| Specific heat, Cv (liquid) | 125.0 | J/mol K | 1.73 | kJ/kg K | 0.414 | Btu/lb°F or cal/g K | |||||
| Ionization potential | 10.34 | eV | |||||||||
| log KOW (Octanol/Water Partition Coefficient) | 3.39 | ||||||||||
| Melting point | 143.48 | K | -129.7 | °C | -201.4 | °F | |||||
| Molecular Weight | 72.149 | g/mol | 0.15906 | lb/mol | |||||||
| Solubility in water, at 25°C | 0.038 | mg/ml | |||||||||
| Sound velocity | 1012 | m/s | 3319 | ft/s | 2267 | mi/h | |||||
| Specific Gravity (gas) (relativ to air) | 2.48 | ||||||||||
| Specific Gravity (liquid) (relativ to water) | 0.63 | ||||||||||
| Specific Heat Ratio (gas) - CP/CV | 1.09 | ||||||||||
| Specific Heat Ratio (liquid) - CP/CV | 1.34 | ||||||||||
| Specific Volume | 0.0001162 | m3/mol | 0.0016106 | m3/kg | 0.8300514 | ft3/slug | 0.0257988 | ft3/lb | |||
| Standard molar entropy, S° (gas) | 348 | J/mol K | 4.82 | kJ/kg K | 1.15 | Btu/lb °F | |||||
| Standard molar entropy, S° (liquid) | 263 | J/mol K | 3.65 | kJ/kg K | 0.87 | Btu/lb °F | |||||
| Surface tension | 16.0 | dynes/cm | 0.016 | N/m | |||||||
| Thermal Conductivity | 0.111 | W/m°C | 0.064135 | Btu/hr ft °F | |||||||
| Triple point pressure | 7.63 ×10-8 | MPa=MN/m2 | 7.63 ×10-7 | bar | 7.53 ×10-7 | atm | 1.11 ×10-5 | psi=lbf/in2 | |||
| Triple point temperature | 143.5 | K | -129.7 | °C | -201.46 | °F | |||||
| Vapor (saturation) pressure | 0.0685 | MPa=MN/m2 | 514.0 | mm Hg | 0.6762 | atm | 9.94 | psi=lbf/in2 | |||
| Viscosity, dynamic (absolute) | 0.2224 | cP | 149.4 | (lbm/ft s ×10-6) | 4.64 | (lbf s/ft2 ×10-6) | |||||
| Viscosity, kinematic | 0.358 | cSt | 3.9 | (ft2/s ×10-6) |
Density of pentane with varying temperature and pressure
See also more about atmospheric pressure, and STP - Standard Temperature and Pressure & NTP - Normal Temperature and Pressure,
as well as Thermophysical properties of: Acetone, Acetylene, Air, Ammonia, Argon, Benzene, Butane, Carbon dioxide, Carbon monoxide, Ethane, Ethanol, Ethylene, Helium, Hydrogen, Hydrogen sulfide, Methane, Methanol, Nitrogen, Oxygen, Propane, Toluene, Water and Heavy water, D2O.
Pentane is a liquid at standard conditions. However, if heated above 36°C/97°F it becomes a gas, and when cooled it becomes a solid. The phase diagram for pentane shows the phase behavior with changes in temperature and pressure. The curve between the critical point and the triple point shows the pentane boiling point with changes in pressure. It also shows the saturation pressure with changes in temperature.
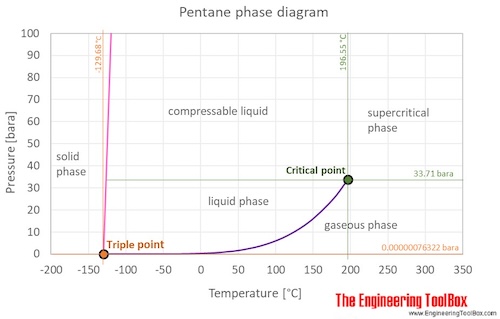
At the critical point there is no change of state when pressure is increased or if heat is added.
The triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.



