Water vs. Steam - Critical and Triple Points
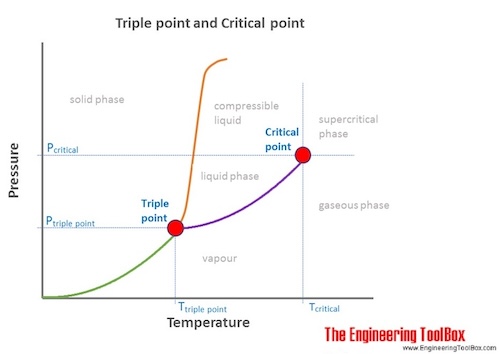
Critical point is where vapor and liquid are indistinguishable and triple point is where ice, water and vapor coexist in thermodynamic equilibrium.
At the critical point there is no change of state when pressure is increased or if heat is added. At the critical point water and steam can't be distinguished and there is no point referring to water or steam.
The critical point of water is achieved at
- Water vapor pressure of 217.75 atm = 220.64 bar = 22.064 MPa = 3200.1 psi
- Temperature of 647.096 K = 373.946 °C = 705.103 °F
- Critical point density: 0.322 g/cm3 = 0.6248 slug/ft3 = 20.102 lbm/ft3
For states above the critical point the steam is supercritical . Supercritical is not the same as superheated - which is saturated steam at lower pressures and temperatures heated above the saturation temperature.
The triple point of a substance is the temperature and pressure at which the three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.
The triple point of water is at
- Water vapor pressure of 0.00604 atm = 6.12 mbar = 611.657 Pa = 0.08871 psi
- Temperature 273.16 K = 0.01 °C = 32.02 °F
- Triple point density (liquid): 0.99979 g/cm3 = 1.93991 slug/ft3 = 62.4148 lbm/ft3
See Water and Heavy Water - for thermodynamic properties.
See also Water Boiling points at high pressure, Boiling points at vacuum pressure, Density, specific weight and thermal expansion coefficient, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pKw , of normal and heavy water, Melting points at high pressure, Saturation pressure, Specific gravity, Specific heat (heat capacity) and Specific volume for online calculatores, figures and tables showing temperature or pressure dependences of the different properties.