Cooling and Heating Equations
Latent and sensible cooling and heating equations - imperial units.
Sensible Heat
The sensible heat in a heating or cooling process of air (heating or cooling capacity) can be calculated in SI-units as
h s = cp ρ q dt (1)
where
h s = sensible heat (kW)
cp = specific heat of air (1.006 kJ/kg oC)
ρ = density of air (1.202 kg/m3 )
q = air volume flow (m3 /s)
dt = temperature difference (oC)
Or in Imperial units as
h s = 1.08 q dt (1b)
where
h s = sensible heat (Btu/hr)
q = air volume flow (cfm, cubic feet per minute)
dt = temperature difference ( oF)
Example - Heating Air, Sensible Heat
Metric Units
An air flow of 1 m3 /s is heated from 0 to 20 oC . Using (1) the sensible heat added to the air can be calculated as
h s = (1.006 kJ/kg oC) (1.202 kg/m3 ) ( 1 m3 /s ) ((20 oC) - (0 oC))
= 24.2 (kW)
Imperial Units
An air flow of 1 cfm is heated from 32 to 52 oF . Using (1b) the sensible heat added to the air can be calculated as
h s = 1.08 (1 cfm) ((52 oF) - (32 oF))
= 21.6 (Btu/hr)
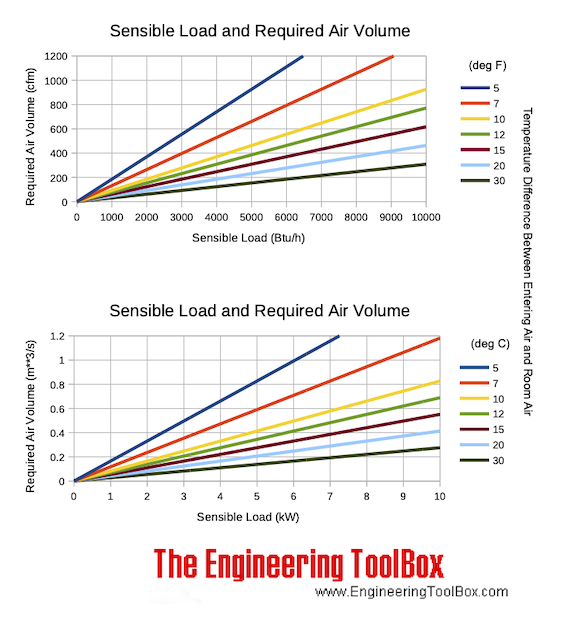
Sensible Heat Load and Required Air Volume Chart
Sensible heat load and required air volume to keep the temperature constant at various temperature differences between make up air and room air:
Latent Heat
Latent heat due to the moisture in air can be calculated in SI-units as:
h l = ρ h we q dw kg (2)
where
h l = latent heat (kW)
ρ = density of air (1.202 kg/m3 )
q = air volume flow (m3 /s)
h we = latent heat evaporization water ( 2454 kJ/kg - in air at atmospheric pressure and 20 oC)
dw kg = humidity ratio difference (kg water/kg dry air)
- estimate humidity with the Mollier diagram
Latent evaporation heat for water can be calculated as
h we = 2494 - 2.2 t (2a)
where
t = evaporation temperature (oC)
Or for Imperial units:
h l = 0.68 q dw gr (2b)
or
h l = 4840 q dw lb (2c)
where
h l = latent heat (Btu/hr)
q = air volume flow (cfm, cubic feet per minute)
dw gr = humidity ratio difference (grains water/lb dry air)
dw lb = humidity ratio difference (lb water/lb dry air)
-
1 grain = 0.000143 lb = 0.0648 g
- Psychrometric chart
Example - Cooling Air, Latent Heat
Metric Units
An air flow of 1 m3 /s is cooled from 30 to 10 oC . The relative humidity of the air is 70% at the start and 100% at the end of the cooling process.
From the Mollier diagram we estimate the water content in the hot air to be 0.0187 kg water/kg dry air, and the water content in the cold air to be 0.0075 kg water/kg dry air .
Using (2) the latent heat removed from the air can be calculated as
h l = (1.202 kg/m3 ) ( 2454 kJ/kg ) ( 1 m3 /s ) (( 0.0187 kg water/kg dry air ) - ( 0.0075 kg water/kg dry air ))
= 34.3 (kW)
Imperial Units
An air flow of 1 cfm is cooled from 52 to 32 oF . The relative humidity of the air is 70% at the start and 100% at the end of the cooling process.
From the psychrometric chart we estimate the water content in the hot air to be 40 grains water/lb dry air, and the water content in the cold air to be 26 grains water/lb dry air .
Using (2b) the latent heat removed from the air can be calculated as
h l = 0.68 (1 cfm) (( 40 grains water/lb dry air ) - ( 26 grains water/lb dry air ))
= 9.5 (Btu/hr)
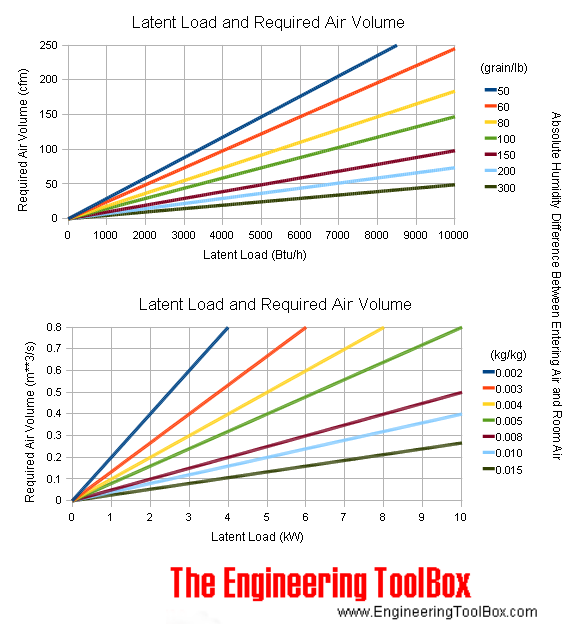
Latent Heat Load and Required Air Volume Chart
Latent heat load - humidifying and dehumidifying - and required air volume to keep temperature constant at various temperature differences between entering air and room air are indicated in the chart below:
Total Heat - Latent and Sensible Heat
Total heat due to both temperature and moisture can be expressed in SI units as:
h t = ρ q dh (3)
where
h t = total heat (kW)
q = air volume flow (m3 /s)
ρ = density of air (1.202 kg/m3 )
dh = enthalpy difference (kJ/kg)
- estimate enthalpy with the Mollier diagram
Or - in imperial units:
h t = 4.7 q dh (3b)
where
h t = total heat (Btu/hr)
q = air volume flow (cfm, cubic feet per minute)
dh = enthalpy difference (btu/lb dry air)
Total heat can also be expressed as:
h t = h s + h l
= 1.08 q dt + 0.68 q dw gr (4)
Example - Cooling or Heating Air, Total Heat
Metric Units
An air flow of 1 m3 /s is cooled from 30 to 10 oC . The relative humidity of the air is 70% at the start and 100% at the end of the cooling process.
From the Mollier diagram we estimate the water enthalpy in the hot air to be 77 kJ/kg dry air, and the enthalpy in the cold air to be 28 kJ/kg dry air .
Using (3) the total sensible and latent heat removed from the air can be calculated as
h t = (1.202 kg/m3 ) ( 1 m3 /s ) (( 77 kJ/kg dry air ) - (28 kJ/kg dry air ))
= 58.9 (kW)
Imperial Units
An air flow of 1 cfm is cooled from 52 to 32 oF . The relative humidity of the air is 70% at the start and 100% at the end of the cooling process.
From the psychrometric chart we estimate the water enthalpy in the hot air to be 18.7 Btu /lb dry air, and the enthalpy in the cold air to be 11.8 Btu /lb dry air .
Using (3b) the total sensible and latent heat removed from the air can be calculated as
h t = 4.7 (1 cfm) (( 18.7 Btu /lb dry air ) - ( 11.8 Btu /lb dry air ))
= 32.4 (Btu/hr)
SHR - Sensible Heat Ratio
The Sensible Heat Ratio can be expressed as
SHR = h s / h t (6)
where
SHR = Sensible Heat Ratio
h s = sensible heat
h t = total heat (sensible and latent)
Related Topics
-
Air Conditioning Systems
Design of Air Conditioning systems - heating, cooling and dehumidification of indoor air for thermal comfort.
Related Documents
-
Air - Heating, Cooling, Mixing, Humidifying or Dehumidifying Processes
Basic air changing state processes - heating, cooling, mixing, humidifying and dehumidifying by adding steam or water - psychometric diagrams and the Mollier charts. -
Air - Humidifying by Adding Steam or Water
Air can be humidified by adding water or steam. -
Air - Humidifying with Steam, SI units
Using steam to humidify air. -
Air Heating Systems
Air heating buildings - heat supply vs. air flow and temperature. -
Arithmetic and Logarithmic Mean Temperature Difference
Arithmetic Mean Temperature Difference in Heat Exchangers - AMTD - and Logarithmic Mean Temperature Difference - LMTD - formulas with examples - Online Mean Temperature Calculator. -
Cooling and Heating - Performance and Efficiency Terminology
Performance and efficiency terminology related to heat pumps and air condition systems. -
Cooling Load - Latent and Sensible Heat
Latent and sensible cooling loads to consider when designing HVAC systems. -
Dehumidifiers
Classification of dehumidifiers. -
Electric Heating of Mass
Electric heating of an object or mass - temperature change vs. energy supplied. -
Great Sensible Heat Factor - GSHF
The Great Sensible Heat Factor is the ratio sensible to total heat in a cooling coil. -
Heat Losses from Electrical Equipment
Heat loss from electrical equipment like switch-gear, transformers and variable frequency drives. -
Humid Air - Heating
Enthalpy change and temperature rise when heating humid air without adding moisture. -
HVAC Abbreviations
Commonly used HVAC abbreviations. -
Latent Heat Flow
Latent heat is the heat when supplied to or removed from air results in a change in moisture content - the temperature of the air is not changed. -
Mixing Liquids and/or Solids - Final Temperatures
Calculate the final temperature when liquids or solids are mixed -
Mixing of Humid Air
The change in state wwhen mixing moist air - enthalpy, heat, temperature and specific humidity. -
Moist Air - the Mollier Diagram
The Mollier diagram is a graphic representation of the relationship between air temperature, moisture content and enthalpy - and is a basic design tool for building engineers and designers. -
Removing Heat with Air
Calculating heat removed with air by measuring the wet bulb temperature. -
Room Sensible Heat Factor - RSHF
Room Sensible Heat Factor - RSHF - is defined as the sensible heat load divided by the total heat load in a room -
Sensible Heat Flow
Sensible heat is dry heat causing change in temperature but not in moisture content -
Sensible Heat Ratio - SHR
The ratio Sensible Heat (or Cooling) Load to the Total Heat (or Cooling) Load. -
Steam Heating Air
Calculate steam heated air systems. -
Steam Heating Processes - Load Calculating
Calculating the amount of steam in non-flow batch and continuous flow heating processes. -
Steam Radiators and Convectors - Heating Capacities
Steam radiators and steam convectors - heating capacities and temperature coefficients.