Mixing of Humid Air
The change in state wwhen mixing moist air - enthalpy, heat, temperature and specific humidity.
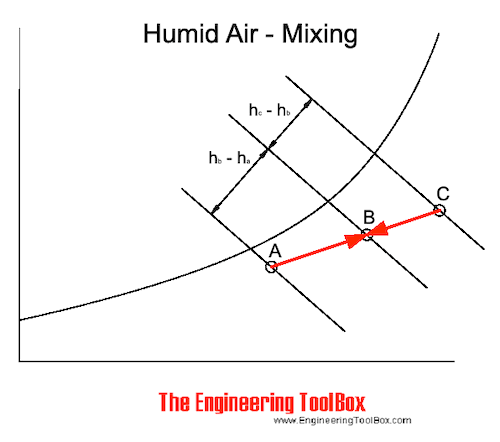
In the Mollier diagram - or in the psychrometric chart - when mixing air of state A and C, the mixing point will be on a straight line in point B.
The position of point B depends on the air volume (or mass) of the air in state A and C.
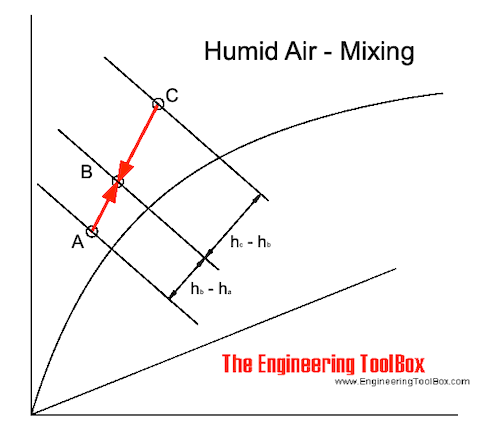
The mixing process are indicated in a Mollier diagram above and in a psychrometric chart below.
Enthalpy Balance when Mixing Air
The heat or enthalphy balance when humid air is mixed can be expressed as:
QA hA + QC hC = (QA + QC) hB (1)
where
Q = volume of the air (m3)
h = enthalpy of the humid air (kJ/m3)
By transforming (1) the enthalpy of the mixed air can be expressed as:
hB = (QA hA + QC hC) / (QA + QC) (1b)
Note! It's common to use air volume when calculating ventilation or air conditioning systems. Using the mass of air is more accurate but within the operation limits of common ventilation systems the volume approach is accurate enough. Be aware that this may not be true in industrial high temperature air handling processes - like dryers or similar.
Moisture Balance when Mixing Air
The moisture balance when humid air is mixed can be expressed as:
QA xA + QC xC = (QA + QC) xB (2)
where
x = specific humidity or humidity ratio (kgwater/kgdry_air)
By transforming (2) - the specific humidity of the mixed air can be expressed as:
xB = (QA xA + QC xC) / (QA + QC) (2b)
Humid Air and Fog
When hot humid air is mixed with cold air the result may be fog.
If the mixing point is below the saturation line - as indicated in the figure below - the moisture in the air condensates to small droplets floating in the air.
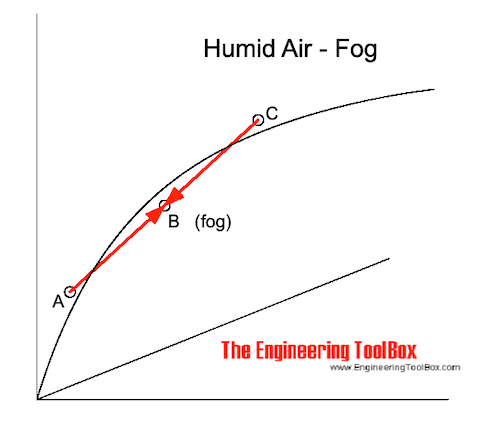
For temperatures below 0 oC the water droplets freezes to ice and snow is created.
Resulting Temperature when Humid Air is Mixed
Mixing Point is above the Saturation Line
As long as the mixed air is above the saturation line (no fog in the mix) the temperature balance can be expressed by modifying the enthalpy balance like
QA cpa tA + QC cpa tC = (QA + QC) cpa tB (3)
where
cpa = 1.01 - specific heat capacity of air at constant pressure (kJ/kgoC, kWs/kgK)
The temperature in the mix can be expressed by transforming (3):
tB = (QA tA + QC tC) / (QA + QC) (3b)
Mixing Point is below Saturation Line
If the mixing point is below the saturation line - water is condensed as droplets and fog is created.
The amount of condense water can be estimated by following the constant enthalpy line from B to the saturation line. The condensate water is the difference between the specific humidity in point B and in the point where the enthalpy line crosses the saturation line.
The mixed temperature is where the enthalpy line crosses the saturation line.
Example - Mixing Humid Air
Mixing Point is above the Saturation Line
1 m3 of air at 25 oC and 50% relative humidity (C) is mixed with 1 m3 air at -5 oC and 80% relative humidity (A).
From the Mollier diagram the humidity ratio of (C) is 0.0097 kg/kg. The enthalpy is 50 kJ/kg. The humidity ratio of (A) is 0.002 kg/kg. The enthalpy is 0 kJ/kg.
The humidity ratio of the mixture can be calculated like
xB= (QA xA + QC xC) / (QA + QC) (4)
= ((1 kg) (0.002 kg/kg) + (1 kg) (0.0097 kg/kg)) / ((1 kg) + (1 kg))
= 0.0058 kg/kg
The enthalpy of the mixed air can be calculated like
hB = ((1 kg) (0 kJ/kg) + (1 kg) (50 kJ/kg)) / ((1 kg) + (1 kg))
= 25 kJ/kg
The temperature of the mixed air can be calculated as:
tB = ((1 kg) (-5 oC) + (1 kg) (25 oC)) / ((1 kg) + (1 kg))
= 10oC
Based on the information above and the Mollier diagram - the relative humidity in the mixing point can be estimated to approximately 80%.
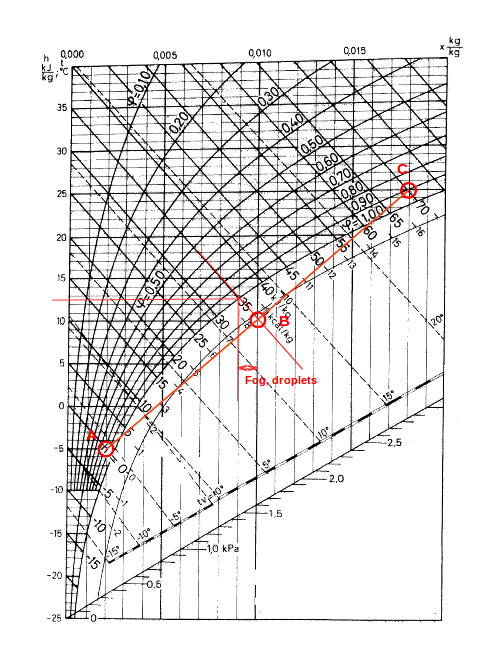
The Mixing Point is below the Saturation Line
1 m3 of air at 25 oC and 90% relative humidity (C) is mixed with 1 m3 air at -5 oC and 80% relative humidity (A).
From the Mollier diagram the humidity ratio of (C) is 0.018 kg/kg. The enthalpy is 70 kJ/kg. The humidity ratio of (A) is 0.002 kg/kg. The enthalpy is 0 kJ/kg.
The enthalpy of the mixture can be calculated as:
hB = ((1 kg) (0 kJ/kg) + (1 kg) (70 kJ/kg)) / ((1 kg) + (1 kg))
= 35 kJ/kg
The humidity ratio of the mixture can be calculated as:
xB = ((1 kg) (0.002 kg/kg) + (1 kg) (0.018 kg/kg)) / ((1 kg) + (1 kg))
= 0.01 kg/kg
Note! This is the humidity ratio including the water droplets - the fog.
The "virtual" mixing point in the Mollier diagram is where the enthalpy line - 35 kJ/kg and the humidity ratio line - 0.01 kg/kg - intersect.
The temperature in the mixed air can be found in the diagram where the enthalpy line - 35 kJ/kg - crosses the saturation line. From the Mollier diagram the mixed temperature is approximately 12.7 oC.
According the Mollier diagram the moisture ratio at this point is approximately 0.0089 kg/kg. The moisture created as fog or droplets is
(0.01 kg/kg) - (0.0089 kg/kg)
= 0.0011 kg/kg