Hydrocarbons, Alcohols and Acids - Boiling points
Boiling temperatures (°C and °F) with varying carbon numbers up to C33.
The tables and figures below show how the boiling point changes with increasing carbon number up to C 33 for different kinds of hydrocarbons, alcohols and carboxylic acids. More detailed definitions and examples of molecular structures of the different groups are given below the figures.
- Melting point - the temperature at which a solid turns into a liquid
- Boiling point - the temperature at which a liquid turns into a gas
For hydrocarbons with the same carbon number the boiling point increases in the following order:
multisubstituted alkane < singelsubstituted alkane < singelsubstituted alkene < normal alkene < normal alkane < alkyl cyclohexane < alkylbenzene < cycloalkene < cycloalkane < 2-, 4- and 3-alkanol / 1-alkylnaphthalene < 1-alkanol < normal alkanoic acid
For melting points, the trends are more varying with increasing carbon number for the different types of hydrocarbons.
See also Melting points of hydrocarbons, alcohols and acids , densities for different kinds of organic compounds and density, boiling and melting points of nitrogen and sulfur compounds.
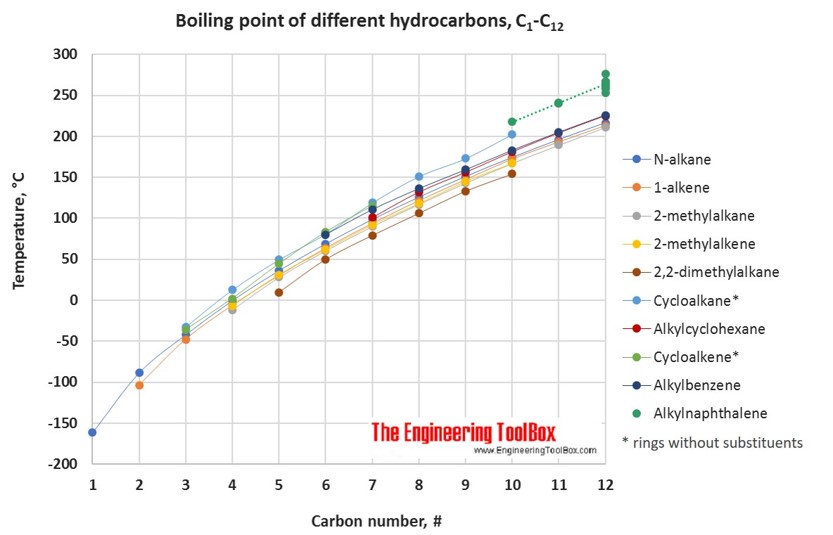
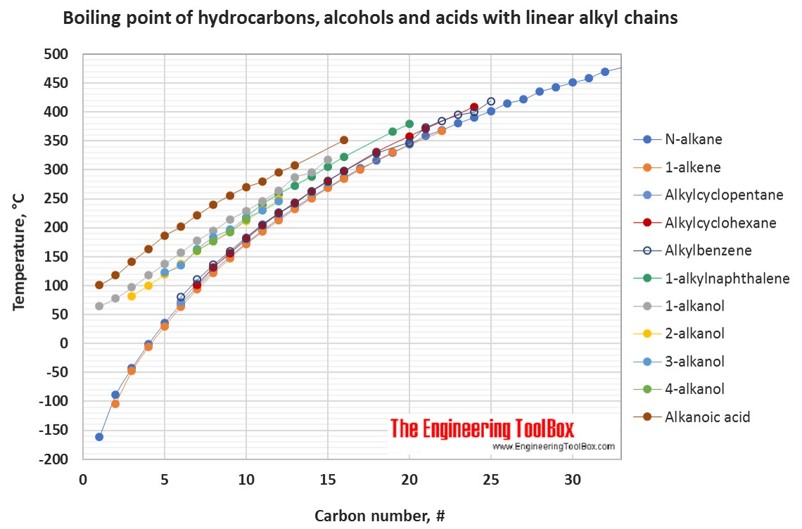
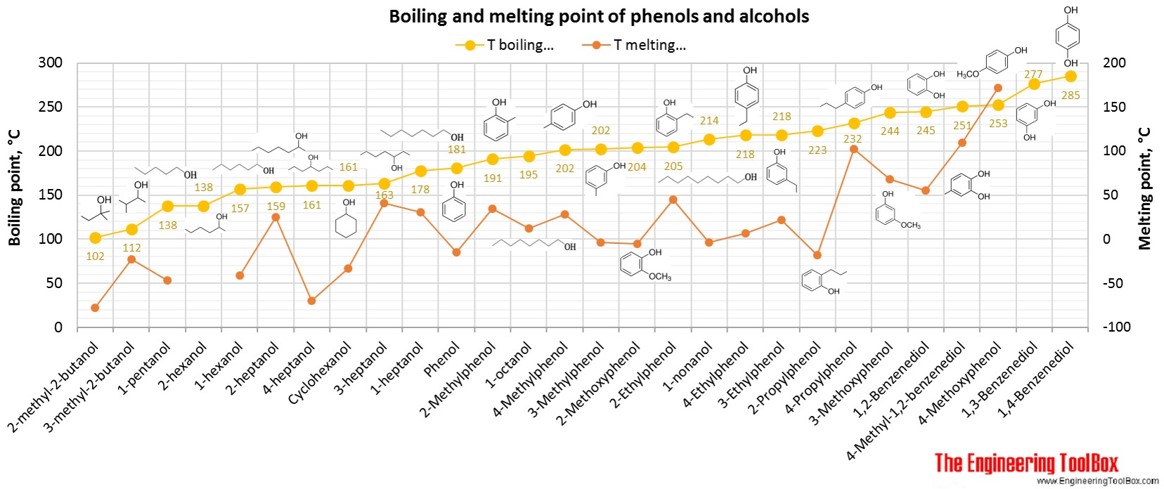
See also pKa values for phenols, alcohols and carboxylic acids .
For full table - rotate the screen!
| Carbon number | Boiling point of hydrocarbons, alcohols and acids, C1-C16, °C | |||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | |
| 2,2-dimetylalkane | 10 | 50 | 79 | 106 | 133 | 155 | ||||||||||
| 2-methylalkane | -12 | 28 | 60 | 90 | 117 | 143 | 167 | 189 | 211 | |||||||
| 2-methylalkene | -7 | 31 | 62 | 92 | 118 | 145 | 167 | |||||||||
| 3-methylalkane | 63 | 92 | 120 | 144 | 168 | 192 | 212 | |||||||||
| 1-alkene | -104 | -48 | -6 | 30 | 64 | 94 | 121 | 147 | 172 | 193 | 213 | 233 | 251 | |||
| N-alkane | -162 | -89 | -42 | -1 | 36 | 69 | 98 | 126 | 151 | 174 | 196 | 216 | 235 | 254 | 270 | 287 |
| 1-alkyne | -84 | -23 | 8 | 40 | 71 | 100 | 126 | 151 | 174 | 196 | 215 | 234 | 284 | |||
| Alkylcyclohexane* | 101 | 132 | 156 | 178 | 204 | 225 | 244 | 263 | ||||||||
| Alkylcyclopentane | 72 | 104 | 131 | 156 | 180 | 206 | 224 | 242 | 262 | 279 | ||||||
| Alkylbenzene* | 80 | 111 | 136 | 159 | 183 | 205 | 226 | 242 | 263 | |||||||
| Cycloalkene** | -36 | 2 | 44 | 83 | 115 | |||||||||||
| Cycloalkane** | -33 | 13 | 49 | 81 | 119 | 151 | 173 | 202 | ||||||||
| 2-alkanol | 82 | 99 | 119 | 138 | 159 | 179 | 194 | 212 | 231 | 249 | ||||||
| 4-alkanol | 161 | 176 | 193 | 214 | ||||||||||||
| 3-alkanol | 123 | 135 | 163 | 184 | 197 | 217 | 230 | 246 | ||||||||
| 1-alkylnaphthalene | 218 | 240 | 258 | 273 | ||||||||||||
| 1-alkanol | 65 | 78 | 97 | 118 | 138 | 157 | 178 | 195 | 214 | 229 | 246 | 264 | 287 | 296 | ||
| Alkanoic acid | 101 | 118 | 142 | 164 | 186 | 202 | 222 | 240 | 256 | 270 | 280 | 296 | 308 | 351 | ||
| Carbon number | Boiling point of hydrocarbons, alcohols and acids, C1-C16, °F | |||||||||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | |
| 2,2-dimetylalkane | 49 | 121 | 174 | 223 | 271 | 311 | ||||||||||
| 2-methylalkane | 11 | 82 | 141 | 194 | 243 | 289 | 333 | 373 | 412 | |||||||
| 2-methylalkene | 20 | 88 | 144 | 198 | 244 | 293 | 333 | |||||||||
| 3-methylalkane | 146 | 198 | 248 | 291 | 334 | 378 | 414 | |||||||||
| 1-alkene | -155 | -54 | 21 | 86 | 146 | 200 | 250 | 297 | 342 | 379 | 415 | 451 | 484 | |||
| N-alkane | -259 | -127 | -44 | 31 | 97 | 156 | 209 | 258 | 303 | 345 | 385 | 421 | 456 | 488 | 518 | 549 |
| 1-alkyne | -119 | -10 | 47 | 104 | 160 | 212 | 259 | 303 | 345 | 385 | 419 | 453 | 543 | |||
| Alkylcyclohexane* | 214 | 270 | 313 | 352 | 399 | 437 | 471 | 505 | ||||||||
| Alkylcyclopentane | 161 | 219 | 268 | 313 | 356 | 403 | 435 | 468 | 504 | 534 | ||||||
| Alkylbenzene* | 176 | 231 | 277 | 319 | 361 | 401 | 439 | 468 | 505 | |||||||
| Cycloalkene** | -33 | 36 | 112 | 181 | 239 | |||||||||||
| Cycloalkane** | -27 | 55 | 121 | 177 | 246 | 304 | 343 | 396 | ||||||||
| 2-alkanol | 180 | 211 | 246 | 280 | 318 | 354 | 380 | 414 | 448 | 480 | ||||||
| 4-alkanol | 322 | 349 | 379 | 417 | ||||||||||||
| 3-alkanol | 253 | 275 | 325 | 363 | 387 | 423 | 445 | 474 | ||||||||
| 1-alkylnaphthalene | 424 | 464 | 496 | 523 | ||||||||||||
| 1-alkanol | 148 | 173 | 207 | 244 | 280 | 314 | 352 | 382 | 417 | 444 | 475 | 507 | 549 | 564 | ||
| Alkanoic acid | 214 | 244 | 287 | 327 | 367 | 396 | 432 | 464 | 493 | 518 | 536 | 565 | 586 | 664 | ||
| * C#(N-alkyl)=0-10 | ||||||||||||||||
| ** rings without substituents | ||||||||||||||||
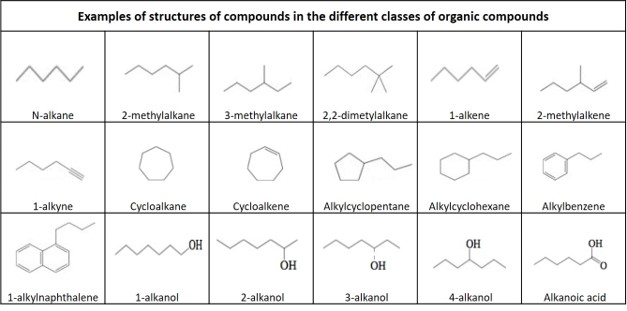
Definitions of organic compounds
Hydrocarbon: An organic compound consisting entirely of hydrogen and carbon.
Main groups of hydrocarbons:
Alkane: An acyclic saturated hydrocarbon, with the general formula C n H 2n+2 . Also called paraffin .
Alkene: An unsaturated hydrocarbon that contains at least one carbon–carbon double bond, with the general formula C n H 2n . Also called olefine .
Alkyne : An unsaturated hydrocarbon containing at least one carbon—carbon triple bond, with the general formula C n H 2n-2 . Also called acetylene .
Cycloalkane: A one-ring (monocyclic) saturated hydrocarbon, with the general formula C n H 2n . Also called naphthene .
Cycloalkene: An alkene hydrocarbon which contains a closed ring of carbon atoms, but has no aromatic character, with the general formula C n H 2n-2 . Also called cycloolefin .
Aromatic hydrocarbon : A cyclic (ring-shaped), planar (flat) molecule with a ring of resonance bonds that exhibits more stability than other geometric or connective arrangements with the same set of atoms. The simplest of the aromatics have 6 carbon atoms and contains 3 double bounds. A one ring aromatic without any substituents is called benzene, with the formula C6 H6.
Polycyclic aromatic hydrocarbons : hydrocarbon that are composed of multiple aromatic rings. A two ring aromatic without any substituents is called naphthalene, with the formula C 10 H 8 .
Some under-groups of hydrocarbons given in this document:
Alkyl: An alkane substituent missing one hydrogen, with general formula C n H 2n+1
2-Methylalkane: A branched alkane, with a methyl group connected to the second carbon atom in the main carbon chain.
3-Methylalkane: A branched alkane, with a methyl group connected to the third carbon atom in the main carbon chain.
2-Methylalkene: A branched alkene, with a methyl group connected to the second carbon atom in the main carbon chain.
Alkylcycklohexane: A monosubstituted cyclohexane with one branching via the attachment of one alkyl group on one carbon of the cyclohexane ring, with the general formula C n H (2n+1) C6 H 11 .
Alkylcycklopentane : A monosubstituted cyclopentane with one branching via the attachment of one alkyl group on one carbon of the cyclohexane ring, with the general formula C n H 2n+1 C5 H 9 .
Alkylbenzene: A monosubstituted benzene with one branching via the attachment of one alkyl group on one carbon of the benzene ring, with the general formula C n H (2n+1) C6 H5.
Alkylnaphthalene: A monosubstituted naphthalene with one branching via the attachment of one alkyl group on one carbon of one of the aromatic rings, with the general formula C n H (2n+1) C 10 H 7 .
Some other groups of organic compounds:
Alcohol: an organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom
Alkanol: An alcohol where the hydroxyl group is connected to an alkane
Carboxylic acid: an organic compound that contains a carboxyl group (C(=O)OH). The general formula of a carboxylic acid is R–COOH, with R referring to the rest of the molecule.
Alkanoic acid: A carboxylic acid where the R is an alkane.
Related Topics
-
Boiling Points of Fluids
Boiling points of elements, products and chemical species at varying conditions. -
Material Properties
Properties of gases, fluids and solids. Densities, specific heats, viscosities and more.
Related Documents
-
Alcohols and Carboxylic Acids - Physical Data
Molweight, melting and boiling point, density, pKa-values, as well as number of carbon and hydrogen atoms in molecules are given for 150 different alcohols and acids. -
Ammonia - Vapour Pressure at Gas-Liquid Equilibrium
Figures and table with ammonia saturation pressure at boiling points, SI and Imperial units. -
Benzene - Thermophysical properties
Chemical, physical and thermal properties of benzene, also called benzol. Phase diagram included. -
Boiling Fluids - Max Suction Flow Velocities
Recommended max suction flow velocity when pumping boiling fluids. -
Crude Oil - Density vs. Temperature
Variations in crude oil density are shown as function of temperatur, together with volume correction factors. -
Crude Oil Properties - Standard test methods (ASTM and others)
An overview of common test methods and typical ranges of variation of petroleum quality parameters. What, why and how do the different test? -
Densities of Aqueous Solutions of Organic Acids
Changes in density of aqueous solutions with changes in concentration at 20°C. Density of acetic acid, citric acid, formic acid, D-lactic acid, oxalic acid and trichloroacetic acid in water is plotted as function of wt%, mol/kg water and mol/l solution. -
Density of Aqueous Solutions of Organic Substances as Sugars and Alcohols
Changes in density of aqueous solutions with changes in concentration at 20°C. Density of some sugars, alcohols and other organic substances in water is plotted as function of wt%, mol/kg water and mol/l solution. -
Elements - Melting Points
Melting points of elements. -
Ethanol - Dynamic and Kinematic Viscosity vs. Temperature and Pressure
Online calculator, figures and tables showing dynamic and kinematic viscosity of ethanol, C2H5OH, at varying temperature and pressure - Imperial and SI Units. -
Ethanol - Density and Specific Weight vs. Temperature and Pressure
Online calculator, figures and tables showing density and specific weight of ethanol at temperatures ranging from -25 to 325 °C (-10 to 620 °F) at atmospheric and higher pressure - Imperial and SI Units. -
Ethanol Freeze Protected Water Solutions
Freezing temperature and flash points for ethanol based water solutions or brines. -
Ethylene - Thermophysical Properties
Chemical, physical and thermal properties of ethylene, also called ethene, acetene and olefiant gas. Phase diagram included. -
Ethylene Glycol Heat-Transfer Fluid Properties
Properties like freezing point, viscosity, specific gravity and specific heat of ethylene glycol based heat-transfer fluids, or brines. -
Fuels - Boiling Points
Fuels and their boiling points. -
Fusion and Evaporation Heat of common Materials
Melting points, heat of fusions, boiling points and heat to evaporate common substances - like hydrogen, water, gold and more .. -
Glycerine - Boiling and Freezing Points
Boiling and freezing points of glycerine aqueous solutions. -
Hydrocarbons - Physical Data
Molweight, melting and boiling point, density, flash point and autoignition temperature, as well as number of carbon and hydrogen atoms in each molecule for 200 different hydrocarbons. -
Jet Fuel - Density vs. Temperature
Variations in jet fuel density as function of temperatur, together with volume correction factors. -
Liquids - Freezing and Melting Points
Common fluids and their freezing and melting points. -
Liquids and Gases - Boiling Points
Boiling temperatures for common liquids and gases - acetone, butane, propane and more. -
Lubricating Oil - Densities vs. Temperature
Variations in lubricating oil density as function of temperatur, together with volume correction factors. -
Melting and Boiling Temperatures - Evaporation and Melting Heats common Materials
Melting and boiling point temperatures, latent heat of evaporation, and melting heat of common substances like copper, gold, lead and more - SI units. -
Melting points of Hydrocarbons, Alcohols and Acids
Melting temperature (°C and °F) with carbon number up to C33. -
Metal Alloys - Melting Points
Alloys and their melting points. -
Metals - Boiling Temperatures
Metals and their boiling temperatures. -
Metals and Alloys - Melting Temperatures
The melting temperatures for some common metals and alloys. -
Methane - Dynamic and Kinematic Viscosity vs. Temperature and Pressure
Online calculator, figures and tables showing dynamic and kinematic viscosity of methane, CH4, at varying temperature and pressure - Imperial and SI Units. -
Methanol - Dynamic and Kinematic Viscosity vs. Temperature and Pressure
Online calculator, figures and tables showing dynamic and kinematic viscosity of liquid methanol,CH3OH, at varying temperature - Imperial and SI Units. -
Molybdenum Binary Eutectic Alloys - Melting Points
Mo - Molybdenum - binary eutectic alloys and melting points. -
Naming of Organic Compounds
Nomenclature rules for different groups of organic compounds and functional groups, together with examples of use of the rules. -
Organic Nitrogen Compounds - Physical Data
Boiling and melting points of amines, diamines, pyrroles, pyridines, piperidines and quinolines shown together with their molecular structures, as well as molweights and density. -
Organic Sulfur Compounds - Physical Data
Boiling and melting points of thoils, sulfides, disulfides and thiophenes shown together with molecular structures, as well as molweights and density. -
Pentane - Density and Specific Weight vs. Temperature and Pressure
Online calculator, figures and table showing density and specific weight of pentane, C5H12, at temperatures ranging from -130 to 325 °C (-200 to 620 °F) at atmospheric and higher pressure - Imperial and SI Units. -
Petroleum Products - Average Boiling Points
Definition, explanation and examples of calculation of various types of average boiling point of petroleum products and other mixtures of hydrocarbons: VABP, MABP, WABP, CABP and MeABP. -
Petroleum Products - Standard Test Methods (ASTM and others) and Specifications
An overview of common test methods and specifications of petroleum fuels. What, why and how do the different test? -
Phenols, Alcohols and Carboxylic Acids - pKa Values
For oxygen containing organic compounds this is given: pKa (the negative logarithm of the acid dissociation constant), molecular structures, molar weights, density and melting and boiling points. -
Propylene Glycol based Heat-Transfer Fluids
Freezing points of propylene glycol based heat-transfer fluids suitable for the food processing industry. -
Refrigerants - Physical Properties
Physical properties of refrigerants - molecular weight, boiling, freezing and critical points. -
Water - Boiling Points at Higher Pressures
Online calculator, figures and tables showing boiling points of water at pressures ranging from 14.7 to 3200 psia (1 to 220 bara). Temperature given as °C, °F, K and °R. -
Water - Boiling Points vs. Altitude
Elevation above sea level and the boiling point of water. -
Yield Structure of Crude Oils with Increasing Density of Crude
Yields of different crude oil distillation cuts are plotted as function of whole crude specific gravity. Fractions based on European and North American markets, and the typical differences in crude oil fractionation in the two markets are also shown.




