Petroleum Products - Average Boiling Points
Definition, explanation and examples of calculation of various types of average boiling point of petroleum products and other mixtures of hydrocarbons: VABP, MABP, WABP, CABP and MeABP.
Boiling point: T he temperature at which a liquid turns into a gas
A mixture of different compounds boils over a certain range of temperature, reflecting the boiling point of each specific compound present in the mixture. For many purposes, it is suitably to calculate an average boiling point (ABP) of mixtures.
Then, the boiling point of each compound is weighted with regard to the fraction of the compound compared to the whole blend. There are several types of fractions that can be calculated: The mole fraction, the weight fraction and the volume fraction, and the type of fraction must be specified when the term average boiling point is used. There is also a cubic average boiling temperature and a mean average boiling temperature . The definitions of each type of average boiling point are given by the formulas:

See also Average boiling point from gravity and molecular weight
Example: "Gasoline" mixture
Gasoline consists of branched paraffins, cycloparaffins and aromatics with carbon numbers typically from C 4 to C 12 .
A "simple" gasoline could then contain a mixture of iso-pentane, 3-methylpentane, iso-octane, meta-xylene and hexylcyclohexane. The table below shows calculations of the different kinds of average boiling points of a mixture containing the given volume fractions of the five compounds
For full table - rotate the screen!
| Compound | Molweight | Density@15°C | Boiling point | X vol | #mol | X mol | Weight | X w |
| g/mol | g/cm3 | °C | cm3 /cm3 | mol | mol/mol | g | g/g | |
| Iso-pentane | 72.2 | 0.620 | 28 | 0.1 | 0.00086 | 0.13 | 0.062 | 0.084 |
| 3-methylpentane | 86.2 | 0.660 | 63 | 0.2 | 0.00153 | 0.23 | 0.132 | 0.180 |
| Iso-octane | 114.2 | 0.690 | 99 | 0.3 | 0.00181 | 0.27 | 0.207 | 0.282 |
| Meta-xylene | 106.2 | 0.860 | 139 | 0.2 | 0.00162 | 0.24 | 0.172 | 0.234 |
| Hexylcyclohexane | 168.3 | 0.808 | 225 | 0.2 | 0.00096 | 0.14 | 0.162 | 0.220 |
| MABP, °C | WABP, °C | VABP, °C | CABP, °C | MeABP, °C | ||||
| 109.3 | 123.6 | 117.9 | 106.5 | 107.9 | ||||
It can be seen that the WABP is biased against the densest components, while the MABP is biast against the lightest components.
However, a real gasoline contains hundreds of different compounds with individual properties, and a similar calculation is not possible to performe due to the complexity and lack of precise analytical tools.
The way to get past this restriction is to use a distillation curve obtained using a real distillation, as ASTM D86 , or a gas chromatographic analysis, as ASTM D2887, to calculate VABP or WABP and then, use agreed correction factors to find the other kinds of ABPs.
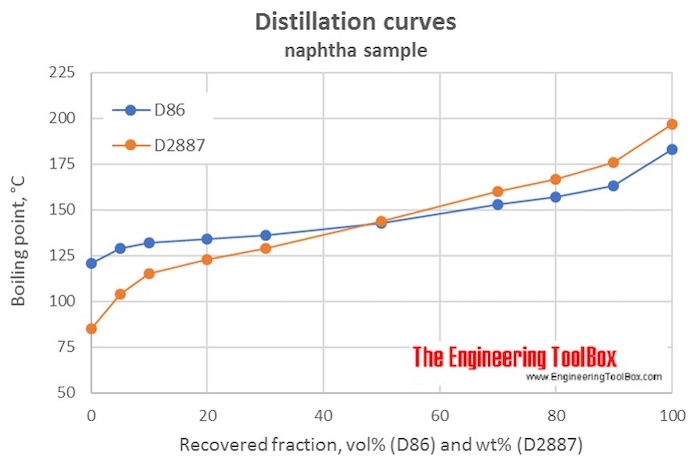
The table below shows results from a D86 and a D2887 analysis of a naphtha sample, and the calculated VABP and WABP and the slopes for the distillation curves, using the formulas:
VABP = 0.2*(T 10% + T 30% + T 50% + T 70% + T 90% ) from D86
WABP = 0.2*(T 10% + T 30% + T 50% + T 70% + T 90% ) from D2887
Slope = (T 90% - T 10% )/80
| Fraction of sample recovered | D86 | D2887 | ||
| Vol% (D86) or Wt% (D2887) | °C | °F | °C | °F |
| IBP | 121 | 85 | ||
| 5 | 129 | 104 | ||
| 10 | 132 | 115 | ||
| 20 | 134 | 123 | ||
| 30 | 136 | 129 | ||
| 50 | 143 | 144 | ||
| 70 | 153 | 160 | ||
| 80 | 157 | 167 | ||
| 90 | 163 | 176 | ||
| FBP | 183 | 197 | ||
| VABP | 145 | 294 | ||
| WABP | 145 | 293 | ||
| Slope, °C/% recovered | 0.39 | 0.76 | ||
| Slope, °F/% recovered | 0.70 | 1.37 | ||
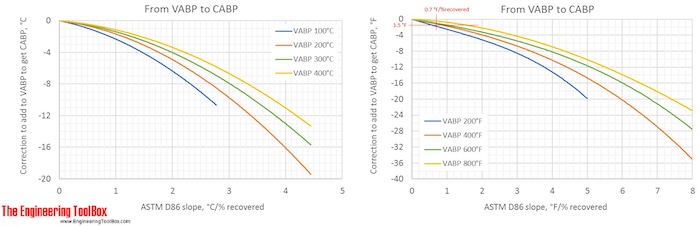
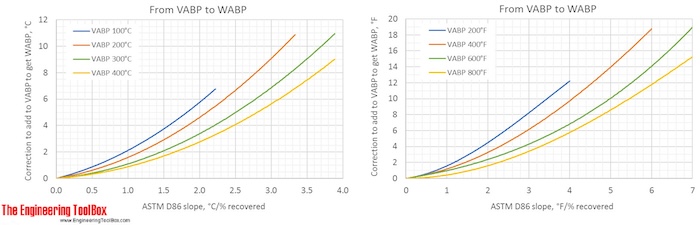
Correction factors are found by using the figures below. Note! The figures are using °C or °F, and results from D86.
The narrower the cut of the oil fraction (smaller slope), the smaller is the correction.
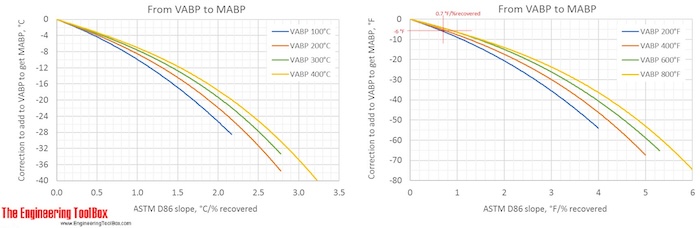
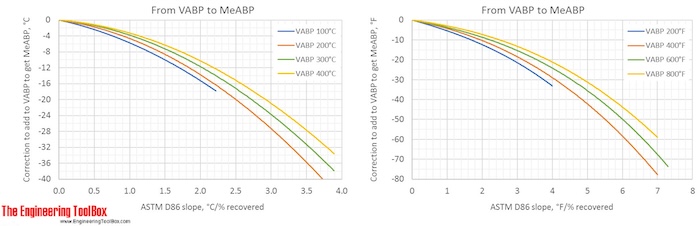
The figures show that the correction from a VABP of 294°F to MABP is -6°F for a sample with a slope of 0.7°F/% recovered. That gives a MABP of 288°F.
The correction from a VABP of 294°F to CABP is only -1.5°F for a sample with a slope of 0.7°F/% recovered. (To see more detailes, right hand click on the figure and choose view image)
Related Topics
-
Boiling Points of Fluids
Boiling points of elements, products and chemical species at varying conditions. -
Fluid Mechanics
The study of fluids - liquids and gases. Involving velocity, pressure, density and temperature as functions of space and time. -
Material Properties
Properties of gases, fluids and solids. Densities, specific heats, viscosities and more.
Related Documents
-
Alcohols and Carboxylic Acids - Physical Data
Molweight, melting and boiling point, density, pKa-values, as well as number of carbon and hydrogen atoms in molecules are given for 150 different alcohols and acids. -
Crude Oil - Density vs. Temperature
Variations in crude oil density are shown as function of temperatur, together with volume correction factors. -
Crude Oil Properties - Standard test methods (ASTM and others)
An overview of common test methods and typical ranges of variation of petroleum quality parameters. What, why and how do the different test? -
Fuel Oils - Densities vs. Temperature
Variations in fuel oils density as function of temperatur, together with volume correction factors. -
Gasoline - Density, Specific Heat, Viscosity and Thermal Conductivity vs. Temperature
Density, specific heat, dynamic and kinematic viscosity and thermal conductivity of gasoline vs. temperature -
Hydrocarbon Mixtures - Average Boiling Points vs. Gravity and Molecular Weights
Formulas and examples of calculation of boiling point of hydrocarbon mixtures from gravity and molecular weight. -
Hydrocarbon Mixtures - Molecular Weight vs. Gravity and Average Boiling Point
Formulas and examples of calculation of average molecular weight of hydrocarbon mixtures from gravity and average boiling point, achieved from distillation data. -
Hydrocarbons - Physical Data
Molweight, melting and boiling point, density, flash point and autoignition temperature, as well as number of carbon and hydrogen atoms in each molecule for 200 different hydrocarbons. -
Hydrocarbons - Melting Point vs. Molecular Weight
Calculate melting point of hydrocarbons from molecular weight (molar mass). -
Hydrocarbons, Alcohols and Acids - Boiling points
Boiling temperatures (°C and °F) with varying carbon numbers up to C33. -
Hydrocarbons, Linear Alcohols and Acids - Densities
Density of hydrocarbons like alcohols and acids as function of carbon number at 20°C / 68°. -
Jet Fuel - Density vs. Temperature
Variations in jet fuel density as function of temperatur, together with volume correction factors. -
Lubricating Oil - Densities vs. Temperature
Variations in lubricating oil density as function of temperatur, together with volume correction factors. -
Organic Nitrogen Compounds - Physical Data
Boiling and melting points of amines, diamines, pyrroles, pyridines, piperidines and quinolines shown together with their molecular structures, as well as molweights and density. -
Organic Sulfur Compounds - Densities
Liquid density of different kinds of organic sulfur compounds with varying carbon number (20°C/68°F). Comparison of thiols, sulfides, disulfides and thiophenes. -
Organic Sulfur Compounds - Physical Data
Boiling and melting points of thoils, sulfides, disulfides and thiophenes shown together with molecular structures, as well as molweights and density. -
Petroleum Products - Standard Test Methods (ASTM and others) and Specifications
An overview of common test methods and specifications of petroleum fuels. What, why and how do the different test? -
Yield Structure of Crude Oils with Increasing Density of Crude
Yields of different crude oil distillation cuts are plotted as function of whole crude specific gravity. Fractions based on European and North American markets, and the typical differences in crude oil fractionation in the two markets are also shown.




