Definitions of the acid dissociation constant and pKa are given below the figures, together with the definition of some classes of organic acids .
In the table below, pKa1 and pKa2 for water solutions at 25°C are given together with boiling and melting point, density and molecular weight , as well as number of carbon, hydrogen and oxygen atoms in each molecule.
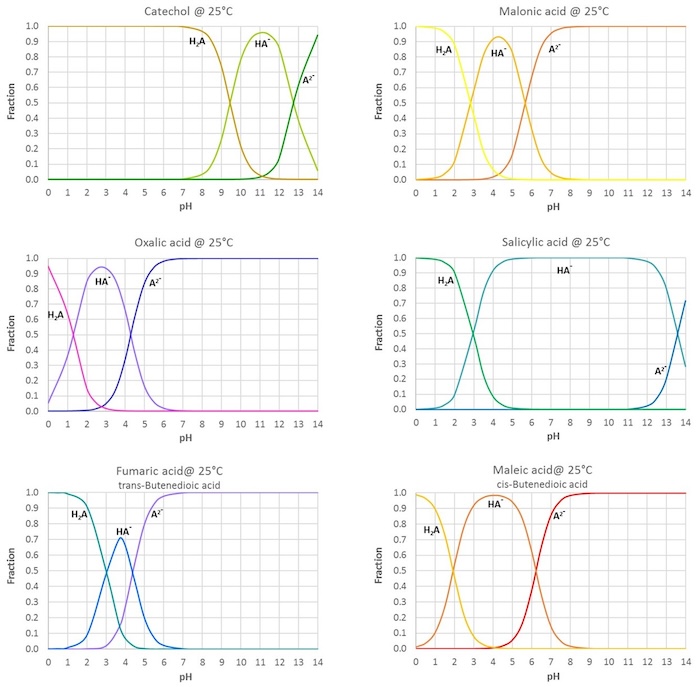
Below the table, figures showing the fractions of the different acid forms in aqueous solutions at varying pH are given for some common diprotic organic acids (values calculated from the tabulated pKa s).
See also boiling and melting point of hydrocarbons, alcohols and acids, pKa of amines, diamines and cyclic organic nitrogen compounds and pKa of inorganic acids and bases, as well as acid base indicators, Acid-base properties of aqueous solutions of salts with ions from both acids and bases and Buffer solutions

For full table with Common Names, Boiling temperatures and Densities - rotate the screen!
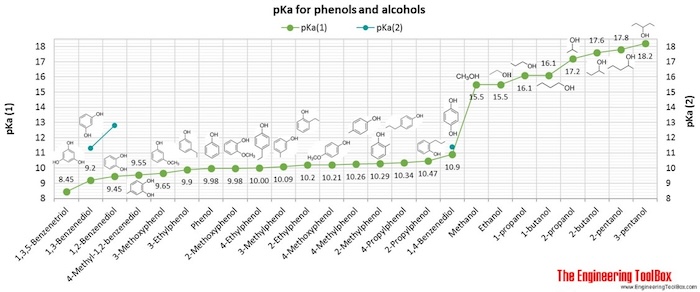
Phenols, Alcohols and Carboxylic Acids - pKa Values
| Group | IUPAC name | Common name | #C | #H | #O | pKa1 | pKa2 | Molweight
g/mol | Melting temp
°C | Boiling temp
°C | Density
@20°C5*
g/ml |
|---|
| 1-alkanol |
Methanol |
Methyl alcohol |
1 |
4 |
1 |
15.5 |
|
32.04 |
-98 |
65 |
0.791 |
| 1-alkanol |
Ethanol |
Ethyl alcohol |
2 |
6 |
1 |
15.5 |
|
46.07 |
-114 |
78 |
0.789 |
| 1-alkanol |
1-propanol |
Propyl alcohol |
3 |
8 |
1 |
16.1 |
|
60.09 |
-124 |
97 |
0.804 |
| 1-alkanol |
1-butanol |
Butyl alcohol |
4 |
10 |
1 |
16.1 |
|
74.12 |
-89 |
118 |
0.810 |
|
|
|
|
|
|
|
|
|
|
|
|
| 2-alkanol |
2-propanol |
Isopropyl alcohol, isopropanol |
3 |
8 |
1 |
17.2 |
|
60.09 |
-88 |
82 |
0.785 |
| 2-alkanol |
2-butanol |
sec-butyl alcohol |
4 |
10 |
1 |
17.6 |
|
74.12 |
-88 |
99 |
0.806 |
| 2-alkanol |
2-pentanol |
sec-amyl alcohol |
5 |
12 |
1 |
17.8 |
|
88.15 |
-73 |
119 |
0.809 |
|
|
|
|
|
|
|
|
|
|
|
|
| 3-alkanol |
3-pentanol |
Diethyl carbinol |
5 |
12 |
1 |
18.2 |
|
88.15 |
-70 |
123 |
0.820 |
|
|
|
|
|
|
|
|
|
|
|
|
| Phenols |
Phenol |
Hydroxybenzene |
6 |
6 |
1 |
9.98 |
|
94.11 |
41 |
181 |
1.132* |
| Phenols |
1,2-Benzenediol |
Catechol, pyrocatechol |
6 |
6 |
2 |
9.45 |
12.8 |
110.11 |
102 |
245 |
1.344* |
| Phenols |
1,3-Benzenediol |
Resorcinol |
6 |
6 |
2 |
9.2 |
11.3 |
110.11 |
110 |
277 |
1.278 |
| Phenols |
1,4-Benzenediol |
Hydroquinone, 1,4-Dihydroxybenzene |
6 |
6 |
2 |
10.9 |
11.4 |
110.11 |
171 |
285 |
|
| Phenols |
1,3,5-Benzenetriol |
Phloroglucinol, 1,3,5-trihydroxybenzene |
6 |
6 |
3 |
8.45 |
|
126.11 |
216 |
s |
1.460* |
| Phenols |
2-Methylphenol |
o-cresol, 2-methylhydroxybenzene |
7 |
8 |
1 |
10.29 |
|
108.13 |
31 |
191 |
1.135* |
| Phenols |
3-Methylphenol |
m-cresol, 3-methylhydroxybenzene |
7 |
8 |
1 |
10.09 |
|
108.13 |
12 |
202 |
1.03* |
| Phenols |
4-Methylphenol |
p-cresol, 4-methylhydroxybenzene |
7 |
8 |
1 |
10.26 |
|
108.13 |
35 |
202 |
1.154* |
| Phenols |
2-Methoxyphenol |
Guaiacol |
7 |
8 |
2 |
9.98 |
|
124.13 |
28 |
204 |
1.1287* |
| Phenols |
3-Methoxyphenol |
Resorcinol, monomethyl ether |
7 |
8 |
2 |
9.65 |
|
124.13 |
-18 |
244 |
1.145* |
| Phenols |
4-Methoxyphenol |
|
7 |
8 |
2 |
10.21 |
|
124.13 |
55 |
253 |
|
| Phenols |
4-Methyl-1,2-benzenediol |
4-Methylcatechol |
7 |
8 |
2 |
9.55 |
|
124.13 |
68 |
251 |
1.129* |
| Phenols |
2-Ethylphenol |
2-Ethylhydroxybenzene |
8 |
10 |
1 |
10.2 |
|
122.16 |
-3 |
205 |
1.015* |
| Phenols |
3-Ethylphenol |
3- Ethylhydroxybenzene |
8 |
10 |
1 |
9.9 |
|
122.16 |
-4 |
218 |
1.008* |
| Phenols |
4-Ethylphenol |
4-Ethylhydroxybenzene |
8 |
10 |
1 |
10.00 |
|
122.16 |
45 |
218 |
1.05* |
| Phenols |
2-Propylphenol |
o-propylphenol |
9 |
12 |
1 |
10.47 |
|
136.19 |
7 |
223 |
1.015 |
| Phenols |
4-Propylphenol |
p-propylphenol |
9 |
12 |
1 |
10.34 |
|
136.19 |
22 |
232 |
1.009* |
|
|
|
|
|
|
|
|
|
|
|
|
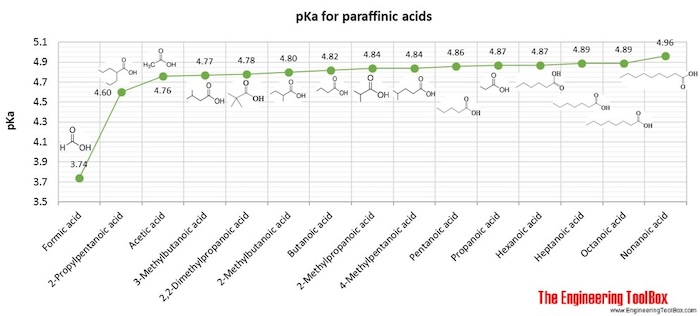
| Alkanoic acid |
Formic acid |
Methanoic acid |
1 |
2 |
2 |
3.74 |
|
46.0 |
8 |
101 |
1.220 |
| Alkanoic acid |
Acetic acid |
Ethanoic acid |
2 |
4 |
2 |
4.76 |
|
60.1 |
17 |
118 |
1.0446* |
| Alkanoic acid |
Butanoic acid |
Butyric acid |
4 |
8 |
2 |
4.82 |
|
88.1 |
-5 |
164 |
0.9528* |
| Alkanoic acid |
Pentanoic acid |
Valeric acid |
5 |
10 |
2 |
4.86 |
|
102.1 |
-34 |
186 |
0.9339* |
| Alkanoic acid |
Propanoic acid |
Propionic acid |
3 |
6 |
2 |
4.87 |
|
74.1 |
-21 |
142 |
0.9882* |
| Alkanoic acid |
Hexanoic acid |
Caproic acid |
6 |
12 |
2 |
4.87 |
|
116.2 |
-4 |
202 |
0.9212* |
| Alkanoic acid |
Heptanoic acid |
Enanthic acid |
7 |
14 |
2 |
4.89 |
|
130.2 |
-7 |
222 |
0.9124* |
| Alkanoic acid |
Octanoic acid |
Caprylic acid |
8 |
16 |
2 |
4.89 |
|
144.2 |
17 |
240 |
0.907* |
| Alkanoic acid |
Nonanoic acid |
Pelargonic acid |
9 |
18 |
2 |
4.96 |
|
158.2 |
12 |
256 |
0.905 |
|
|
|
|
|
|
|
|
|
|
|
|
| Alkanoic acid, branched |
2-Methylpropanoic acid |
Isobutyric acid |
4 |
8 |
2 |
4.84 |
|
88.1 |
-46 |
155 |
0.943* |
| Alkanoic acid, branched |
2,2-Dimethylpropanoic acid |
Trimethylacetic acid |
5 |
10 |
2 |
4.78 |
|
102.1 |
35 |
164 |
0.9052* |
| Alkanoic acid, branched |
2-Methylbutanoic acid |
|
5 |
10 |
2 |
4.80 |
|
102.1 |
-70 |
176 |
0.934 |
| Alkanoic acid, branched |
3-Methylbutanoic acid |
Isovaleric acid |
5 |
10 |
2 |
4.77 |
|
102.1 |
-30 |
176 |
0.925 |
| Alkanoic acid, branched |
4-Methylpentanoic acid |
4-Methylvaleric acid, Isocaproic acid |
6 |
12 |
2 |
4.84 |
|
116.2 |
-33 |
200 |
0.923 |
| Alkanoic acid, branched |
2-Propylpentanoic acid |
Valproic acid |
8 |
16 |
2 |
4.60 |
|
144.2 |
|
223 |
0.904* |
|
|
|
|
|
|
|
|
|
|
|
|
| Phenylalkanoic acid |
Phenylethanoic acid |
α-Tolylic acid, Benzeneacetic |
8 |
8 |
2 |
4.31 |
|
136.1 |
77 |
266 |
1.081* |
| Phenylalkanoic acid |
2-Phenylbutyric acid |
a-Ethyl-a-toluic acid |
10 |
12 |
2 |
4.66 |
|
47.5 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
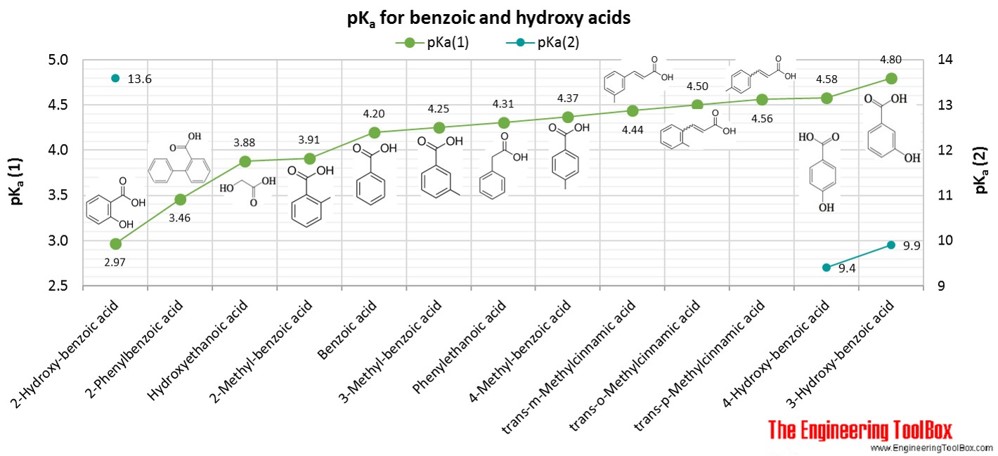
| Benzoic acid |
Benzoic acid |
Benzenecarboxylic acid |
7 |
6 |
2 |
4.20 |
|
122.1 |
122 |
249 |
1.2663* |
| Benzoic acid |
2-Methyl-benzoic acid |
o-Toluic acid |
8 |
8 |
2 |
3.91 |
|
136.1 |
107 |
258 |
1.0624* |
| Benzoic acid |
3-Methyl-benzoic acid |
m-Toluic acid |
8 |
8 |
2 |
4.25 |
|
136.1 |
111 |
|
1.0544* |
| Benzoic acid |
4-Methyl-benzoic acid |
p-Toluic acid |
8 |
8 |
2 |
4.37 |
|
136.1 |
182 |
275 |
|
| Benzoic acid |
2-Phenylbenzoic acid |
|
13 |
10 |
2 |
3.46 |
|
198.2 |
112 |
344 |
|
|
|
|
|
|
|
|
|
|
|
|
|
| Cinnamic acid |
trans-m-Methylcinnamic acid |
|
10 |
10 |
2 |
4.44 |
|
|
115 |
|
|
| Cinnamic acid |
trans-o-Methylcinnamic acid |
|
10 |
10 |
2 |
4.50 |
|
162.2 |
175 |
|
|
| Cinnamic acid |
trans-p-Methylcinnamic acid |
|
10 |
10 |
2 |
4.56 |
|
|
199 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Hydroxy acid |
Hydroxyethanoic acid |
Glycolic acid |
2 |
4 |
3 |
3.88 |
|
76.1 |
80 |
d |
|
| Hydroxy acid |
2-Hydroxy-benzoic acid |
Salicylic acid |
7 |
6 |
3 |
2.97 |
13.6 |
138.1 |
159 |
|
1.443 |
| Hydroxy acid |
3-Hydroxy-benzoic acid |
|
7 |
6 |
3 |
4.80 |
9.9 |
138.1 |
202 |
|
|
| Hydroxy acid |
4-hydroxy-benzoic acid |
|
7 |
6 |
3 |
4.58 |
9.4 |
138.1 |
215 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
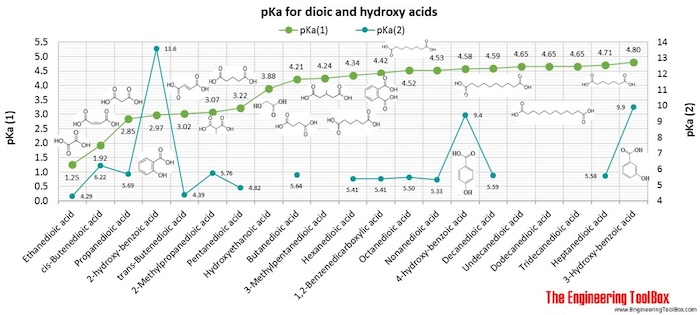
| Dioic acid |
Ethanedioic acid |
Oxalic acid |
2 |
2 |
4 |
1.25 |
4.29 |
90.0 |
d 190 |
s 157 |
1.93* |
| Dioic acid |
Propanedioic acid |
Malonic acid |
3 |
4 |
4 |
2.85 |
5.69 |
104.1 |
136 |
d 140 |
1.6193* |
| Dioic acid |
cis-Butenedioic acid |
Maleic |
4 |
4 |
4 |
1.92 |
6.22 |
116.1 |
139 |
d |
1.59* |
| Dioic acid |
trans-Butenedioic acid |
Fumaric |
4 |
4 |
4 |
3.02 |
4.39 |
116.1 |
s 300 |
|
1.635 |
| Dioic acid |
2-Methylpropanedioic acid |
Methylmalonic acid |
4 |
6 |
4 |
3.07 |
5.76 |
118.1 |
129 |
|
1.455 |
| Dioic acid |
Butanedioic acid |
Succinic acid |
4 |
6 |
4 |
4.21 |
5.64 |
118.1 |
185 |
d 235 |
1.572* |
| Dioic acid |
Pentanedioic acid |
Glutaric acid |
5 |
8 |
4 |
3.22 |
4.82 |
132.1 |
98 |
273 |
1.4293* |
| Dioic acid |
3-Methylpentanedioic acid |
3-Methylglutaric acid |
6 |
10 |
4 |
4.24 |
|
146.1 |
83 |
|
|
| Dioic acid |
Hexanedioic acid |
Adipic acid |
6 |
10 |
4 |
4.34 |
5.41 |
146.1 |
152 |
338 |
|
| Dioic acid |
Heptanedioic acid |
Pimelic acid |
7 |
12 |
4 |
4.71 |
5.58 |
160.2 |
104 |
357 d? |
|
| Dioic acid |
1,2-Benzenedicarboxylic acid |
o-Phthalic |
8 |
6 |
4 |
4.42 |
5.41 |
166.1 |
d 210 |
|
|
| Dioic acid |
Octanedioic acid |
Suberic acid |
8 |
14 |
4 |
4.52 |
5.50 |
174.2 |
142 |
230 d? |
|
| Dioic acid |
Nonanedioic acid |
Azelaic acid |
9 |
16 |
4 |
4.53 |
5.33 |
188.2 |
110 |
336 d? |
|
| Dioic acid |
Decanedioic acid |
Sebacic acid |
10 |
18 |
4 |
4.59 |
5.59 |
202.2 |
131 |
374 d? |
1.2705 |
| Dioic acid |
Undecanedioic acid |
1,9-Nonanedicarboxylic acid |
11 |
20 |
4 |
4.65 |
|
216.3 |
109 |
|
|
| Dioic acid |
Dodecanedioic acid |
Decane-1,10-dicarboxylic acid |
12 |
22 |
4 |
4.65 |
|
230.3 |
128 |
417 d? |
|
| Dioic acid |
Tridecanedioic acid |
Brassylic acid |
13 |
24 |
4 |
4.65 |
|
244.3 |
113 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Chlorocarboxylic acid |
Chloroacetic acid |
CH2ClCO2H |
2 |
3 |
2 |
2.87 |
|
94.5 |
62 |
189 |
1.40435* |
| Chlorocarboxylic acid |
Dichloroacetic acid |
CHCl2CO2H |
2 |
2 |
2 |
1.35 |
|
128.9 |
12 |
193 |
1.564 |
| Chlorocarboxylic acid |
Trichloroacetic acid |
CCl3CO2H |
2 |
1 |
2 |
0.66 |
|
163.4 |
59 |
198 |
1.612* |
| Chlorocarboxylic acid |
2-Chloropropanoic acid |
C2H4ClCO2H |
3 |
5 |
2 |
2.83 |
|
108.5 |
|
185 |
1.26 |
| Chlorocarboxylic acid |
3-Chloropropanoic acid |
C2H4ClCO2H |
3 |
5 |
2 |
3.98 |
|
108.5 |
|
204 d |
|
| Chlorocarboxylic acid |
2-Chlorobutanoic acid |
C3H6ClCO2H |
4 |
7 |
2 |
2.86 |
|
122.6 |
|
|
1.18 |
| Chlorocarboxylic acid |
3-Chlorobutanoic acid |
C3H6ClCO2H |
4 |
7 |
2 |
4.05 |
|
122.6 |
16 |
|
1.19 |
| Chlorocarboxylic acid |
4-Chlorobutanoic acid |
C3H6ClCO2H |
4 |
7 |
2 |
4.52 |
|
122.6 |
16 |
|
1.22 |
| Chlorocarboxylic acid |
2-Chlorobenzoic acid |
C6H4ClCO2H |
7 |
5 |
2 |
2.90 |
|
156.6 |
140 |
274 |
1.5445* |
| Chlorocarboxylic acid |
3-Chlorobenzoic acid |
C6H4ClCO2H |
7 |
5 |
2 |
3.84 |
|
156.6 |
154 |
283 |
1.4965* |
| Chlorocarboxylic acid |
4-Chlorobenzoic acid |
C6H4ClCO2H |
7 |
5 |
2 |
4.00 |
|
156.6 |
240 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Bromocarboxylic acid |
Bromoacetic acid |
CH2BrCO2H |
2 |
3 |
2 |
2.90 |
|
139.0 |
50 |
208 |
1.93352* |
| Bromocarboxylic acid |
3-Bromopropanoic acid |
C2H4BrCO2H |
3 |
5 |
2 |
4.00 |
|
153.0 |
63 |
|
1.485* |
| Bromocarboxylic acid |
2-Bromobenzoic acid |
C6H4BrCO2H |
7 |
5 |
2 |
2.85 |
|
201.0 |
149 |
295 |
1.9295* |
| Bromocarboxylic acid |
3-Bromobenzoic acid |
C6H4BrCO2H |
7 |
5 |
2 |
3.81 |
|
201.0 |
157 |
285 |
1.8455* |
| Bromocarboxylic acid |
4-Bromobenzoic acid |
C6H4BrCO2H |
7 |
5 |
2 |
3.96 |
|
201.0 |
254 |
|
1.8945* |
|
|
|
|
|
|
|
|
|
|
|
|
| Fluorocarboxylic acid |
Fluoroacetic acid |
CH2FCO2H |
2 |
3 |
2 |
2.59 |
|
78.1 |
35 |
168 |
1.3682* |
| Fluorocarboxylic acid |
Trifluoroacetic acid |
CF3CO2H |
2 |
1 |
2 |
0.52 |
|
114.0 |
-15 |
72 |
1.5351* |
| Fluorocarboxylic acid |
2-Fluorobenzoic acid |
C6H4FCO2H |
7 |
5 |
2 |
3.27 |
|
140.1 |
124 |
|
1.465* |
| Fluorocarboxylic acid |
3-Fluorobenzoic acid |
C6H4FCO2H |
7 |
5 |
2 |
3.86 |
|
140.1 |
124 |
|
0.475* |
| Fluorocarboxylic acid |
4-Fluorobenzoic acid |
C6H4FCO2H |
7 |
5 |
2 |
4.15 |
|
140.1 |
184 |
|
1.485* |
| Fluorocarboxylic acid |
Pentafluorobenzoic acid |
C6F5CO2H |
7 |
1 |
2 |
1.75 |
|
212.1 |
103 |
220 |
|
s = sublimes, d = decomposes, * measured at 25°C,2* measured at 50°C,3* measured at 15°C,4* measured at 115°C,5* Measured at solid state
d? = Boiling temperature at 1 atm is estimated from low pressure measurements. The decomposition temperature is not known. |
An acid dissociation constant, Ka , is a quantitative measure of the strength of an acid in solution. It is the equilibrium constant for a chemical reaction known as dissociation of acid–base reactions. In aqueous solution, the equilibrium of acid dissociation can be written symbolically as:
HA + H2O = A-+H3 O+
where HA is an acid that dissociates into A-, (known as the conjugate base of the acid) and a hydrogen ion which combines with a water molecule to make a hydronium ion.
The chemical species HA, A-and H3 O+are said to be in equilibrium when their concentrations do not change with the passing of time. The dissociation constant is usually written as a quotient of the equilibrium concentrations (in mol/L), denoted by (HA), (A-) and (H3 O+)
Ka = (A-)*(H3 O+) / (HA)*(H2O)
In all, but the most concentrated, aqueous solutions of an acid the concentration of water can be taken as constant and can be ignored. The definition can then be written more simply
HA = A-+ H+and Ka = (A-)*(H+) / (HA)
This is the definition in common usage. For many practical purposes it is more convenient to discuss the logarithmic constant, pKa
pKa = -log10 Ka
The larger the value of pKa , the smaller the extent of dissociation at any given pH - that is, the weaker the acid.
A weak acid has a pKa value in the approximate range -2 to 12 in water.
Strong acids has pKa values of less than about -2; the dissociation of a strong acid is effectively complete such that concentration of the undissociated acid is too small to be measured. pKa values for strong acids can, however, be estimated by theoretical means.
After rearranging the expression defining Ka , and putting pH = -log10 (H+), one obtains
pH = pKa + (A-) / (HA) and further
pH - pKa = log(A-) / (HA)
Then, a solution with 50% dissociation has pH equal to the pKa of the acid.
Polyprotic acids are acids that can lose more than one proton. Then we have more than one dissiciation constant; Ka1 , Ka2 , etc.. and similar pKa1 , pKa2 , etc.
All data given in the figures apply to dilute aqueous solutions at ambient temperature.
The figure below shows the fractions of the different acid forms (H2A, HA-and A2- for twoprotic acids) in aqueous solutions at varying pH :
Definition of organic compounds
Hydrocarbon: An organic compound consisting entirely of hydrogen and carbon.
Alkane: An acyclic saturated hydrocarbon, with the general formula Cn H2n+2 . Also called paraffin.
Aromatic hydrocarbon : A cyclic (ring-shaped), planar (flat) molecule with a ring of resonance bonds that exhibits more stability than other geometric or connective arrangements with the same set of atoms. The simplest of the aromatics have 6 carbon atoms and contains 3 double bounds. A one ring aromatic without any substituents is called benzene, with the formula C6H6.
Alkyl: An alkyl group is an alkane substituent missing one hydrogen, with general formula Cn H2n+1.
Phenyl: An phenyl group is a benzene substituent missing one hydrogen, with general formula C6 H5.
Alcohol: An organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom.
Alkanol: An alcohol where the hydroxyl group is bound to an alkyl group, with the general formula Cn H2n+1 OH.
Phenol: An alcohol where the hydroxyl group is bound to a phenyl group, with the formula C6H5OH.
Phenols : A group of compounds consisting of a phenol molecule with one or more substiuents.
Diol: An organic compound containing two hydroxyl groups, -OH.
Triol: An organic compound containing three hydroxyl groups, -OH.
Carboxylic acid: an organic compound that contains a carboxyl group (C(=O)OH). The general formula of a carboxylic acid is R–COOH, with R referring to the rest of the molecule.
Paraffinic acid = Alkanoic acid: A carboxylic acid where the R is an alkyl group.
Dioic acid: A carboxylic acid with two acid groups (-COOH).
Benzoic acid: A carboxylic acid where the acid group is substituted to one carbon of a benzene ring.
Hydroxy acid: An carboxylic acid containing an additional hydroxy group (-OH), substistuted to another carbon atom than the acid group.









