Water Density, Specific Weight and Thermal Expansion Coefficients - Temperature and Pressure Dependence
Data on the density and specific weight of water across various temperatures and pressures. Useful for engineering, fluid dynamics, and HVAC calculations.
Density is the ratio of the mass to the volume of a substance:
ρ = m / V (1)
where
ρ = density, units typically (g/cm3) or (lb/ft3)
m = mass, units typically (g) or (lb)
V = volume, units typically (cm3) or (ft3)
Pure water has its highest density 1000 kg/m3 or 1.940 slug/ft3 at temperature 4°C (=39.2°F).
Specific weight is the ratio of the weight to the volume of a substance:
γ = (m g) / V
= ρ g (2)
where
γ = specific weight, units typically (N/m3) or (lbf/ft3)
m = mass, units typically (g) or (lb)
g = acceleration due to gravity, units typically (m/s2) and value on Earth usually given as 9.80665 m/s2 or 32.17405 ft/s2
V = volume, units typically (cm3) or (ft3)
ρ = density, units typically (g/cm3) or (lb/ft3)
Example 1: Specific weight of water
In the SI system, specific weight of water at 4°C will be:
γ = 1000 (kg/m3) 9.807 (m/s2) = 9807 (kg/(m2 s2)) = 9807 (N/m3) = 9.807 (kN/m3)
In the Imperial system the mass unit is the slug (sl) , and is derived from the pound-force by defining it as the mass that will accelerate at 1 foot per square second when a 1 pound-force acts upon it :
1 (lbf) = 1 (sl) 1 (ft/s2) and 1 (sl) = 1 (lbf)/1 (ft/s2)
The density of water is 1.940 sl/ft3 at 39 °F (4 °C), and the specific weight in Imperial units is
γ = 1.940 (sl/ft3) 32.174 (ft/s2) = 1.940 (lbf)/((ft/s2) (ft3)) * 32.174 (ft/s2) = 62.4 (lbf/ft3)
See more about the difference between mass and weight
Online Water density Calculator
The calculator below can be used to calculate the liquid water density at given temperatures.
The output density is given as g/cm3, kg/m3, lb/ft3, lb/gal(US liq) and sl/ft3.
Note! Temperature must be within the ranges 0-370 °C, 32-700 °F, 273-645 K and 492-1160 °R to get valid values.
The density of water depends on temperature and pressure as shown below:
See Water and Heavy Water for thermodynamic properties at standard condtions.
See also other properties of Water at varying temperature and pressure : Boiling points at high pressure, Boiling points at vacuum pressure, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pKw, of normal and heavy water, Melting points at high pressure, Prandtl number, Properties at Gas-Liquid Equilibrium Conditions, Saturation pressure, Specific gravity, Specific heat (heat capacity), Specific volume, Thermal conductivity, Thermal diffusivity and Vapour pressure at gas-liquid equilibrium .
For other substances, see density and specific weight of acetone, air, ammonia, argon, benzene, butane, carbon dioxide, carbon monoxide, ethane, ethanol, ethylene, helium, hydrogen, methane, methanol, nitrogen, oxygen, pentane, propane and toluene .
Density of crude oil , Density of fuel oils , Density of lubricating oil and Density of jet fuel as function of temperature.
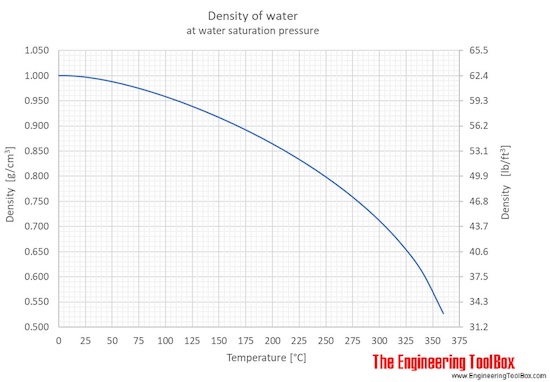
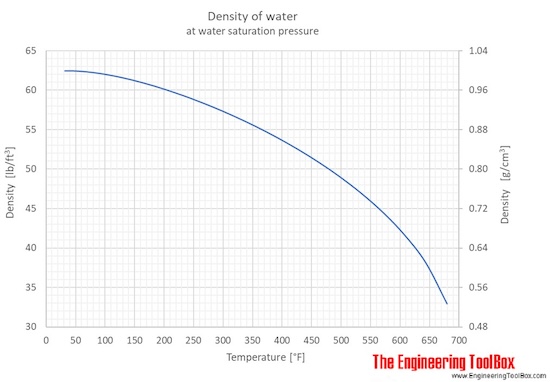
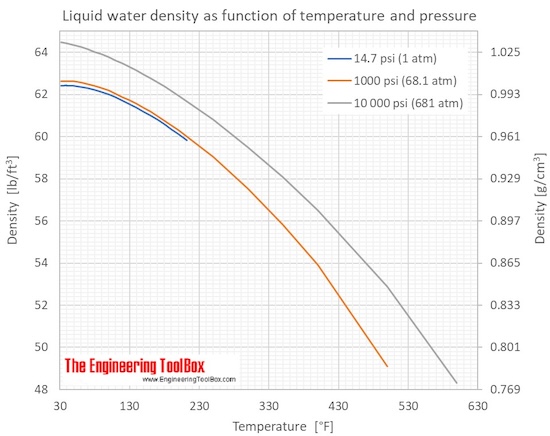
As shown in the figures, the change in density is not linear with temperature - this means that the volumetric expansion coefficient for water is not constant over the temperature range.
Water density, specific weight and thermal expansion coefficient at temperatures given in degree Celcius:
For full table with Specific Weight and Thermal Expansion Coefficient - rotate the screen!
| Temperature | Density (0-100°C at 1 atm, >100 °C at saturation pressure) | Specific weight | Thermal expansion coefficient | |||||
|---|---|---|---|---|---|---|---|---|
| (°C) | (g/cm3) | (kg/m3) | (sl/ft3) | (lbm/ft3) | (lbm/gal(US liq)) | (kN/m3) | (lbf/ft3) | (*10- 4K-1) |
| 0.1 | 0.9998495 | 999.85 | 1.9400 | 62.4186 | 8.3441 | 9.8052 | 62.419 | -0.68 |
| 1 | 0.9999017 | 999.90 | 1.9401 | 62.4218 | 8.3446 | 9.8057 | 62.422 | -0.50 |
| 4 | 0.9999749 | 999.97 | 1.9403 | 62.4264 | 8.3452 | 9.8064 | 62.426 | 0.003 |
| 10 | 0.9997000 | 999.70 | 1.9397 | 62.4094 | 8.3429 | 9.8037 | 62.409 | 0.88 |
| 15 | 0.9991026 | 999.10 | 1.9386 | 62.3719 | 8.3379 | 9.7978 | 62.372 | 1.51 |
| 20 | 0.9982067 | 998.21 | 1.9368 | 62.3160 | 8.3304 | 9.7891 | 62.316 | 2.07 |
| 25 | 0.9970470 | 997.05 | 1.9346 | 62.2436 | 8.3208 | 9.7777 | 62.244 | 2.57 |
| 30 | 0.9956488 | 995.65 | 1.9319 | 62.1563 | 8.3091 | 9.7640 | 62.156 | 3.03 |
| 35 | 0.9940326 | 994.03 | 1.9287 | 62.0554 | 8.2956 | 9.7481 | 62.055 | 3.45 |
| 40 | 0.9922152 | 992.22 | 1.9252 | 61.9420 | 8.2804 | 9.7303 | 61.942 | 3.84 |
| 45 | 0.99021 | 990.21 | 1.9213 | 61.8168 | 8.2637 | 9.7106 | 61.817 | 4.20 |
| 50 | 0.98804 | 988.04 | 1.9171 | 61.6813 | 8.2456 | 9.6894 | 61.681 | 4.54 |
| 55 | 0.98569 | 985.69 | 1.9126 | 61.5346 | 8.2260 | 9.6663 | 61.535 | 4.86 |
| 60 | 0.98320 | 983.20 | 1.9077 | 61.3792 | 8.2052 | 9.6419 | 61.379 | 5.16 |
| 65 | 0.98055 | 980.55 | 1.9026 | 61.2137 | 8.1831 | 9.6159 | 61.214 | 5.44 |
| 70 | 0.97776 | 977.76 | 1.8972 | 61.0396 | 8.1598 | 9.5886 | 61.040 | 5.71 |
| 75 | 0.97484 | 974.84 | 1.8915 | 60.8573 | 8.1354 | 9.5599 | 60.857 | 5.97 |
| 80 | 0.97179 | 971.79 | 1.8856 | 60.6669 | 8.1100 | 9.5300 | 60.667 | 6.21 |
| 85 | 0.96861 | 968.61 | 1.8794 | 60.4683 | 8.0834 | 9.4988 | 60.468 | 6.44 |
| 90 | 0.96531 | 965.31 | 1.8730 | 60.2623 | 8.0559 | 9.4665 | 60.262 | 6.66 |
| 95 | 0.96189 | 961.89 | 1.8664 | 60.0488 | 8.0274 | 9.4329 | 60.049 | 6.87 |
| 100 | 0.95835 | 958.35 | 1.8595 | 59.8278 | 7.9978 | 9.3982 | 59.828 | 7.03 |
| 110 | 0.95095 | 950.95 | 1.8451 | 59.3659 | 7.9361 | 9.3256 | 59.366 | 8.01 |
| 120 | 0.94311 | 943.11 | 1.8299 | 58.8764 | 7.8706 | 9.2487 | 58.876 | 8.60 |
| 140 | 0.92613 | 926.13 | 1.7970 | 57.8164 | 7.7289 | 9.0822 | 57.816 | 9.75 |
| 160 | 0.90745 | 907.45 | 1.7607 | 56.6503 | 7.5730 | 8.8990 | 56.650 | 11.0 |
| 180 | 0.88700 | 887.00 | 1.7211 | 55.3736 | 7.4024 | 8.6985 | 55.374 | 12.3 |
| 200 | 0.86466 | 864.66 | 1.6777 | 53.9790 | 7.2159 | 8.4794 | 53.979 | 13.9 |
| 220 | 0.84022 | 840.22 | 1.6303 | 52.4532 | 7.0120 | 8.2397 | 52.453 | 16.0 |
| 240 | 0.81337 | 813.37 | 1.5782 | 50.7770 | 6.7879 | 7.9764 | 50.777 | 18.6 |
| 260 | 0.78363 | 783.63 | 1.5205 | 48.9204 | 6.5397 | 7.6848 | 48.920 | 22.1 |
| 280 | 0.75028 | 750.28 | 1.4558 | 46.8385 | 6.2614 | 7.3577 | 46.838 | |
| 300 | 0.71214 | 712.14 | 1.3818 | 44.4574 | 5.9431 | 6.9837 | 44.457 | |
| 320 | 0.66709 | 667.09 | 1.2944 | 41.6451 | 5.5671 | 6.5419 | 41.645 | |
| 340 | 0.61067 | 610.67 | 1.1849 | 38.1229 | 5.0963 | 5.9886 | 38.123 | |
| 360 | 0.52759 | 527.59 | 1.0237 | 32.9364 | 4.4030 | 5.1739 | 32.936 | |
| 373.946 | 0.3220 | 322.0 | 0.625 | 20.102 | 2.6872 | 3.1577 | 20.102 | |
Water density, specific weight and thermal expansion coefficient at temperatures given in degree Fahrenheit:
For full table with Specific Weight and Thermal Expansion Coefficient - rotate the screen!
| Temperature | Density (0-212°F at 1 atm, >212 °F at saturation pressure) | Specific weight | Thermal expansion coefficient | |||||
|---|---|---|---|---|---|---|---|---|
| (°F) | (lb/ft3) | (sl/ft3) | (lbm/gal(US liq)) | (g/cm3) | (kg/m3) | (lbf/ft3) | (kN/m3) | (*10- 4K-1) |
| 32.2 | 62.42 | 1.9400 | 8.3441 | 0.99985 | 999.9 | 62.42 | 9.805 | -0.68 |
| 34 | 62.42 | 1.9402 | 8.3448 | 0.99993 | 999.9 | 62.42 | 9.806 | -0.50 |
| 39.2 | 62.43 | 1.9403 | 8.3452 | 0.99997 | 1000.0 | 62.43 | 9.806 | 0.0031 |
| 40 | 62.42 | 1.9402 | 8.3450 | 0.99995 | 1000.0 | 62.42 | 9.806 | 0.01 |
| 50 | 62.41 | 1.9397 | 8.3429 | 0.99970 | 999.7 | 62.41 | 9.804 | 0.88 |
| 60 | 62.36 | 1.9383 | 8.3369 | 0.99898 | 999.0 | 62.36 | 9.797 | 1.59 |
| 70 | 62.30 | 1.9364 | 8.3283 | 0.99796 | 998.0 | 62.30 | 9.787 | 2.18 |
| 80 | 62.22 | 1.9338 | 8.3172 | 0.99662 | 996.6 | 62.22 | 9.773 | 2.72 |
| 90 | 62.11 | 1.9306 | 8.3035 | 0.99498 | 995.0 | 62.11 | 9.757 | 3.21 |
| 100 | 62.00 | 1.9269 | 8.2877 | 0.99308 | 993.1 | 62.00 | 9.739 | 3.66 |
| 110 | 61.86 | 1.9227 | 8.2697 | 0.99093 | 990.9 | 61.86 | 9.718 | 4.08 |
| 120 | 61.71 | 1.9181 | 8.2499 | 0.98855 | 988.6 | 61.71 | 9.694 | 4.46 |
| 130 | 61.55 | 1.9131 | 8.2283 | 0.98597 | 986.0 | 61.55 | 9.669 | 4.81 |
| 140 | 61.38 | 1.908 | 8.205 | 0.9832 | 983.2 | 61.38 | 9.642 | 5.16 |
| 150 | 61.19 | 1.902 | 8.180 | 0.9802 | 980.2 | 61.19 | 9.613 | 5.47 |
| 160 | 61.00 | 1.896 | 8.154 | 0.9771 | 977.1 | 61.00 | 9.582 | 5.71 |
| 170 | 60.79 | 1.890 | 8.127 | 0.9738 | 973.8 | 60.79 | 9.550 | 6.05 |
| 180 | 60.58 | 1.883 | 8.098 | 0.9704 | 970.4 | 60.58 | 9.516 | 6.31 |
| 190 | 60.35 | 1.876 | 8.068 | 0.9668 | 966.8 | 60.35 | 9.481 | 6.57 |
| 200 | 60.12 | 1.869 | 8.037 | 0.9630 | 963.0 | 60.12 | 9.444 | 6.79 |
| 212 | 59.83 | 1.860 | 7.998 | 0.9584 | 958.4 | 59.83 | 9.398 | 7.07 |
| 220 | 59.63 | 1.853 | 7.971 | 0.9552 | 955.2 | 59.63 | 9.367 | |
| 240 | 59.10 | 1.837 | 7.900 | 0.9467 | 946.7 | 59.10 | 9.283 | |
| 260 | 58.53 | 1.819 | 7.824 | 0.9375 | 937.5 | 58.53 | 9.194 | |
| 280 | 57.93 | 1.800 | 7.744 | 0.9279 | 927.9 | 57.93 | 9.100 | |
| 300 | 57.29 | 1.781 | 7.659 | 0.9177 | 917.7 | 57.29 | 8.999 | |
| 350 | 55.59 | 1.728 | 7.431 | 0.8905 | 890.5 | 55.59 | 8.733 | |
| 400 | 53.67 | 1.668 | 7.175 | 0.8598 | 859.8 | 53.67 | 8.432 | |
| 450 | 51.45 | 1.599 | 6.878 | 0.8242 | 824.2 | 51.45 | 8.083 | |
| 500 | 48.92 | 1.521 | 6.540 | 0.7836 | 783.6 | 48.92 | 7.685 | |
| 550 | 45.95 | 1.428 | 6.142 | 0.7360 | 736.0 | 45.95 | 7.218 | |
| 600 | 42.36 | 1.317 | 5.663 | 0.6786 | 678.6 | 42.36 | 6.655 | |
| 625 | 40.12 | 1.247 | 5.363 | 0.6426 | 642.6 | 40.12 | 6.302 | |
| 650 | 37.35 | 1.161 | 4.993 | 0.5982 | 598.2 | 37.35 | 5.867 | |
| 675 | 33.79 | 1.050 | 4.517 | 0.5412 | 541.2 | 33.79 | 5.307 | |
Water density and specific weight at 1000 psi and given temperatures:
For full table with Specific Weight - rotate the screen!
| Temperature | Density (at 1000 psi or 68.1 atm) | Specific weight | ||||||
|---|---|---|---|---|---|---|---|---|
| (°C) | (°F) | (g/cm3) | (kg/m3) | (sl/ft3) | (lbm/ft3) | (lbm/gal(US liq)) | (lbf/ft3) | (kN/m3) |
| 0.0 | 32 | 1.0031 | 1003.1 | 1.946 | 62.62 | 8.371 | 62.62 | 9.837 |
| 4.4 | 40 | 1.0031 | 1003.1 | 1.946 | 62.62 | 8.371 | 62.62 | 9.837 |
| 10.0 | 50 | 1.0031 | 1003.1 | 1.946 | 62.62 | 8.371 | 62.62 | 9.837 |
| 15.6 | 60 | 1.0024 | 1002.4 | 1.945 | 62.58 | 8.366 | 62.58 | 9.831 |
| 21.1 | 70 | 1.0012 | 1001.2 | 1.943 | 62.50 | 8.355 | 62.50 | 9.818 |
| 26.7 | 80 | 0.9999 | 999.9 | 1.940 | 62.42 | 8.344 | 62.42 | 9.805 |
| 32.2 | 90 | 0.9981 | 998.1 | 1.937 | 62.31 | 8.330 | 62.31 | 9.788 |
| 37.8 | 100 | 0.9962 | 996.2 | 1.933 | 62.19 | 8.314 | 62.19 | 9.769 |
| 43.3 | 110 | 0.9944 | 994.4 | 1.928 | 62.03 | 8.292 | 62.03 | 9.744 |
| 48.9 | 120 | 0.9912 | 991.2 | 1.923 | 61.88 | 8.272 | 61.88 | 9.721 |
| 54.4 | 130 | 0.9888 | 988.8 | 1.919 | 61.73 | 8.252 | 61.73 | 9.697 |
| 60.0 | 140 | 0.9864 | 986.4 | 1.914 | 61.58 | 8.232 | 61.58 | 9.673 |
| 65.6 | 150 | 0.9834 | 983.4 | 1.908 | 61.39 | 8.207 | 61.39 | 9.644 |
| 71.1 | 160 | 0.9803 | 980.3 | 1.902 | 61.20 | 8.181 | 61.20 | 9.614 |
| 76.7 | 170 | 0.9768 | 976.8 | 1.895 | 60.98 | 8.152 | 60.98 | 9.579 |
| 82.2 | 180 | 0.9731 | 973.1 | 1.888 | 60.75 | 8.121 | 60.75 | 9.543 |
| 87.8 | 190 | 0.9696 | 969.6 | 1.881 | 60.53 | 8.092 | 60.53 | 9.509 |
| 93.3 | 200 | 0.9661 | 966.1 | 1.874 | 60.31 | 8.062 | 60.31 | 9.474 |
| 121.1 | 250 | 0.9456 | 945.6 | 1.835 | 59.03 | 7.891 | 59.03 | 9.273 |
| 148.9 | 300 | 0.9217 | 921.7 | 1.788 | 57.54 | 7.692 | 57.54 | 9.039 |
| 176.7 | 350 | 0.8943 | 894.3 | 1.735 | 55.83 | 7.463 | 55.83 | 8.770 |
| 204.4 | 400 | 0.8636 | 863.6 | 1.676 | 53.91 | 7.207 | 53.91 | 8.469 |
| 260.0 | 500 | 0.7867 | 786.7 | 1.526 | 49.11 | 6.565 | 49.11 | 7.715 |
| 284.8 | 544.58 | boiling point | ||||||
Water density and specific weight at 10 000 psi and given temperatures:
For full table with Specific Weight - rotate the screen!
| Temperature | Density (at 10 000 psi or 681 atm) | Specific weight | ||||||
|---|---|---|---|---|---|---|---|---|
| (°C) | (°F) | (g/cm3) | (kg/m3) | (sl/ft3) | (lbm/ft3) | (lbm/gal(US liq)) | (lbf/ft3) | (kN/m3) |
| 0.0 | 32 | 1.033 | 1033 | 2.004 | 64.5 | 8.62 | 64.5 | 10.13 |
| 4.4 | 40 | 1.032 | 1032 | 2.003 | 64.4 | 8.61 | 64.4 | 10.12 |
| 10.0 | 50 | 1.031 | 1031 | 2.000 | 64.4 | 8.60 | 64.4 | 10.11 |
| 15.6 | 60 | 1.029 | 1029 | 1.997 | 64.3 | 8.59 | 64.3 | 10.09 |
| 21.1 | 70 | 1.028 | 1028 | 1.994 | 64.1 | 8.58 | 64.1 | 10.08 |
| 26.7 | 80 | 1.026 | 1026 | 1.990 | 64.0 | 8.56 | 64.0 | 10.06 |
| 32.2 | 90 | 1.024 | 1024 | 1.986 | 63.9 | 8.54 | 63.9 | 10.04 |
| 37.8 | 100 | 1.021 | 1021 | 1.982 | 63.8 | 8.52 | 63.8 | 10.02 |
| 43.3 | 110 | 1.019 | 1019 | 1.977 | 63.6 | 8.51 | 63.6 | 9.99 |
| 48.9 | 120 | 1.017 | 1017 | 1.973 | 63.5 | 8.48 | 63.5 | 9.97 |
| 54.4 | 130 | 1.014 | 1014 | 1.968 | 63.3 | 8.46 | 63.3 | 9.94 |
| 60.0 | 140 | 1.011 | 1011 | 1.962 | 63.1 | 8.44 | 63.1 | 9.92 |
| 65.6 | 150 | 1.008 | 1008 | 1.957 | 63.0 | 8.42 | 63.0 | 9.89 |
| 71.1 | 160 | 1.005 | 1005 | 1.951 | 62.8 | 8.39 | 62.8 | 9.86 |
| 76.7 | 170 | 1.002 | 1002 | 1.945 | 62.6 | 8.37 | 62.6 | 9.83 |
| 82.2 | 180 | 0.999 | 999 | 1.939 | 62.4 | 8.34 | 62.4 | 9.80 |
| 87.8 | 190 | 0.996 | 996 | 1.932 | 62.2 | 8.31 | 62.2 | 9.77 |
| 93.3 | 200 | 0.992 | 992 | 1.926 | 62.0 | 8.28 | 62.0 | 9.73 |
| 121.1 | 250 | 0.974 | 974 | 1.890 | 60.8 | 8.13 | 60.8 | 9.55 |
| 148.9 | 300 | 0.953 | 953 | 1.849 | 59.5 | 7.95 | 59.5 | 9.35 |
| 176.7 | 350 | 0.930 | 930 | 1.805 | 58.1 | 7.76 | 58.1 | 9.12 |
| 204.4 | 400 | 0.905 | 905 | 1.756 | 56.5 | 7.55 | 56.5 | 8.88 |
| 260.0 | 500 | 0.847 | 847 | 1.643 | 52.9 | 7.07 | 52.9 | 8.31 |
| 315.6 | 600 | 0.774 | 774 | 1.501 | 48.3 | 6.46 | 48.3 | 7.59 |
Weight per U.S. gallon is based on 7.48 gallons per cubic foot .
- 1 gal(US liq) = 3.7854 l = 0.8327 gal(UK) = 0.8594 gal(US dry) = 0.1074 bu(US dry) = 0.4297 pk(US dry) = 4 qt(US liq) = 8pt(US liq) = 16 c(US) = 32 gi(US liq) = 128 fl oz(US) = 1024 fl dr(US) = 3.7854×10-3m3= 0.1337 ft3= 4.951×10-3yd3
To convert from density in kg/m3 to other density units - or between units - use the converting values below:
-
1 kg/m3= 1 g/l = 0.001 kg/l = 0.000001 kg/cm3= 0.001 g/cm3= 0.99885 oz/ft3= 0.0005780 oz/in3= 0.16036 oz/gal(UK) = 0.1335 oz/gal(US liq) = 0.06243 lb/ft3= 3.6127x10-5 lb/in3= 1.6856 lb/yd3 = 0.010022 lb/gal(UK) = 0.008345 lb/gal(US liq) = 0.00194032 sl/ft3= 0.0007525 ton(long)/yd3= 0.0008428 ton(short)/yd3
See also density converter
Example 2: Water Density in unit oz/in3
Water density at temperature 20 oC is 998.21 kg/m3(table above). The density in units oz/in3 can with the converting value from above be calculated to
998.21 (kg/m3) * 0.0005780 ((oz/in3)/(kg/m3)) = 0.5797 (oz/in3)
Example 3: Mass of Hot Water
A tank with volume 10 m3contains hot water with temperature 190°F. From the table above the density of water at 190°F is 966.8 kg/m3. The total mass of the water in the tank can be calculated
10 (m3) * 966.8 (kg/m3) = 9668 (kg)
See also hydrostatic pressure in water and energy stored in hot water
Related Searches
water density • specific weight of water • water properties • fluid mechanics • engineering reference • water temperature effects • density table • specific gravity • hydrostatics • fluid dynamics •



