Heat Loss from Open Water Tanks
Due to evaporation the heat loss from an open water tank like a swimming pool may be considerable.
The overall heat loss from an open water tank can be expressed as:
Q = Q evaporation fluid + Q radiation fluid + Q transmission through walls (1)
The heat loss due to evaporation of water from a surface of an open tank is totally dominant at higher water temperatures. In general heat loss through insulated walls can be almost neglected. Major heat loss can be avoided by covering the surface.
Note! - take care with the values below - they are rough estimates and depends on air velocity, air turbulence and air moisture over the surface, water stir and more. How fast a cup of coffee or similar cools down outside with some wind compared inside illustrates the power of these parameters and the roughness of the estimates below.
| Water Temperature ( oF) | Heat Loss from Liquid Surface (Btu/ft2hr) | Heat Loss Through Tank Walls (Btu/ft2hr) | |||||
|---|---|---|---|---|---|---|---|
| Evaporation Loss | Radiation Loss | Total | Bare Steel No Insulation | Insulated (inches) | |||
| 1 | 2 | 3 | |||||
| 90 | 80 | 50 | 130 | 50 | 12 | 5 | 4 |
| 100 | 160 | 70 | 230 | 70 | 15 | 8 | 6 |
| 110 | 240 | 90 | 330 | 90 | 19 | 10 | 7 |
| 120 | 360 | 110 | 470 | 110 | 23 | 12 | 9 |
| 130 | 480 | 135 | 615 | 135 | 27 | 14 | 10 |
| 140 | 660 | 160 | 820 | 160 | 31 | 16 | 12 |
| 150 | 860 | 180 | 1040 | 180 | 34 | 18 | 13 |
| 160 | 1100 | 210 | 1310 | 210 | 38 | 21 | 15 |
| 170 | 1380 | 235 | 1615 | 235 | 42 | 23 | 16 |
| 180 | 1740 | 260 | 2000 | 260 | 46 | 25 | 17 |
| 190 | 2160 | 290 | 2450 | 290 | 50 | 27 | 19 |
| 200 | 2680 | 320 | 3000 | 320 | 53 | 29 | 20 |
| 210 | 3240 | 360 | 3600 | 360 | 57 | 31 | 22 |
- the values above are for 60 oF (15.6 oC) still ambient air
Example - Heat Loss from an open Water Tank
The heat loss from an open water tank with
- water temperature: 150 oF
- surface area: 10 ft2
- un-insulated bare steel tank walls area: 50 ft2
can be estimated with values from the table above as:
Q ( evaporation fluid + radiation fluid) = (1040 Btu/(ft2hr)) (10 ft2)
= 10400 Btu/hr
Q transmission through walls = (180 Btu/(ft2hr)) (50 ft2)
= 9000 Btu/hr
Q = Q ( evaporation fluid + radiation fluid) + Q transmission through walls
= (10400 Btu/hr) + (9000 Btu/hr)
= 19400 Btu/hr
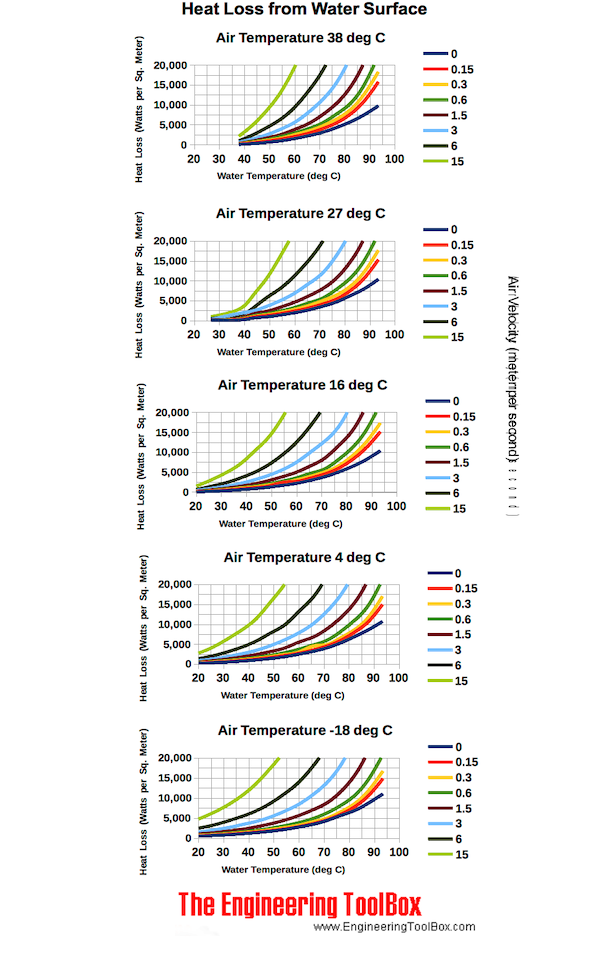
Heat Loss from Water Surfaces Chart - Metric Units
Heat loss are for open tanks in 60 oF still ambient air.
Heat Loss from Water Surfaces Chart - Imperial Units
- T(oC) = 5/9[T( oF) - 32]
- 1 Btu/(ft2hr) = 3.1525 W/m2
Related Topics
-
Heating Systems
Design of heating systems - capacities and design of boilers, pipelines, heat exchangers, expansion systems and more.
Related Documents
-
Content in Horizontal - or Sloped - Cylindrical Tanks or Pipes
Volume of partly filled horizontal or sloped cylindrical tanks or pipes - online calculator. -
Cylindrical Tanks - Volumes
Volume in US gallons and liters. -
Evaporation from a Water Surface
Evaporation of water from a water surface - like a swimming pool or an open tank - depends on water temperature, air temperature, air humidity and air velocity above the water surface - online calculator. -
Evaporative Cooling
Evaporative cooling tutorial. -
Heat Loss from Oil Filled Tanks and Pipe Lines
Heat loss from insulated and non insulated sheltered and exposed oil tanks and pipes. -
Oil Filled Tanks - Heat Loss
Heat loss from insulated and uninsulated, sheltered and exposed heated oil tanks. -
Oil Tanks - Heat Loss
Heat loss from lagged and unlagged, sheltered and exposed oil tanks. -
Pipes Submerged in Water - Heat Emission
Heat emision from steam or water heating pipes submerged in water - assisted (forced) or natural circulation. -
Rectangular Tanks - Volumes
Tank volume per foot depth.