Water-Steam Mollier Diagram
Enthalpy-entropy diagram for water and steam.
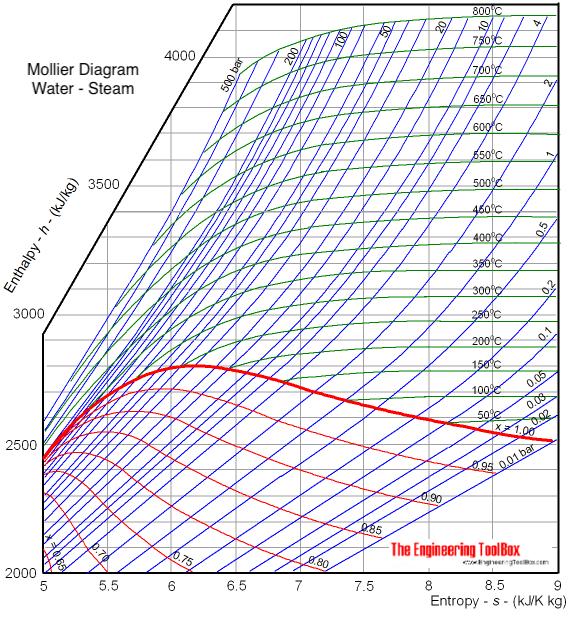
The diagram below can be used to determine enthalpy versus entropy of water and steam.
Download and print Mollier Diagram for Water - Steam
The Mollier diagram is useful when analyzing the performance of adiabatic steady-flow processes, such as flow in nozzles, diffusers, turbines and compressors.
See also Water - Enthalpy (H) and Entropy (S) for figures and tabulated values at varying temperatures
Dryness Fraction
The quantity of saturated vapor in unit mass of wet vapor , denoted by x , is referred to as the
dryness fraction , or quality,
of the vapor.
Convert to other Units
- 1 bar = 105 Pa (N/m2) = 0.1 N/mm2= 10,197 kp/m2= 10.20 m H2O = 0.9869 atm = 14.50 psi (lbf /in2) = 106 dyn/cm2= 750 mmHg
- 1 kJ/kg = 0.4299 Btu/ lbm = 0.23884 kcal/kg
Example - Enthalpy
The enthalpy in saturated steam ( x = 1, the bold red line in the diagram) with pressure 10 bar can be estimated to aprox.
2770 kJ/kg
The value can be verified by using the steam table .
Related Topics
-
Steam and Condensate
Design of steam & condensate systems with properties, capacities, sizing of pipe lines, system configuration and more. -
Thermodynamics
Calculate heat, work, temperature and energy. The thermodynamics of steam and condensate systems. Water and Ice properties. -
Thermodynamics
Work, heat and energy systems.
Related Documents
-
1st Law of Thermodynamics
The First Law of Thermodynamics simply states that energy can be neither created nor destroyed (conservation of energy). Thus power generation processes and energy sources actually involve conversion of energy from one form to another, rather than creation of energy from nothing. -
2nd Law of Thermodynamics
Entropy and disorder. -
Ice - Thermal Properties
Thermal and thermodynamic properties of ice like density, thermal conductivity and specific heat at temperatures from 0 to -100 oC. -
Moist Air - Enthalpy
Sensible and latent heat of moist air. -
Properties of Saturated Steam - SI Units
Saturated Steam Table with steam properties as specific volume, density, specific enthalpy and specific entropy. -
Saturated Steam - Properties - Imperial Units
Steam table with sensible, latent and total heat, and specific volume at different gauge pressures and temperatures. -
Saturated Steam - Properties for Pressure in Bar
Saturated Steam Table with properties like boiling point, specific volume, density, specific enthalpy, specific heat and latent heat of vaporization. -
Steam and Vapor Enthalpy
Vapor and steam enthalpy, specific enthalpy of saturated liquid, saturated vapor and superheated vapor. -
Steam Entropy
Basic steam thermodynamics - entropy diagram. -
Superheated Steam - Enthalpy
Enthalpy of steam superheated to temperatures above it's boiling point. -
Universal and Individual Gas Constants
The Universal and Individual Gas Constants in fluid mechanics and thermodynamics. Individual gas constants for the most common gases. -
Water - Enthalpy and Entropy vs. Temperature
Figures and tables showing the enthalpy and entropy of liquid water as function of temperature - SI and Imperial Units. -
Water - Specific Volume vs. Temperature
Online calculator, figures and tables showing Specific Volume of water at temperatures ranging from 0-370 °C and 32 - 700 °F - Imperial and IS Units. -
Water - Thermophysical Properties
Thermal properties of water at different temperatures like density, freezing temperature, boiling temperature, latent heat of melting, latent heat of evaporation, critical temperature and more. -
Water Vapor - Specific Heat vs. Temperature
Specific heat of Water Vapor - H2O - at temperatures ranging 175 - 6000 K. -
Water vs. Steam - Critical and Triple Points
Critical point is where vapor and liquid are indistinguishable and triple point is where ice, water and vapor coexist in thermodynamic equilibrium. -
Wet Steam - Enthalpy
Wet steam, dryness fraction and enthalpy. -
Wet Steam - Quality vs. Dryness Fractions
Introduction and definition of steam quality and dryness fraction including calculating wet steam enthalpy and specific volume. -
Wet Steam - Specific Volume
Wet steam and specific volume.




