Air - SCFM versus ACFM and ICFM
Actual air compressor capacity (ACFM) vs. standard air capacity (SCFM) and inlet air capacity (ICFM).
SCFM - Standard Cubic Feet per Minute
It is common to rate compressed air consumption in Standard Cubic Feet per Minute - SCFM.
The SCFM - Standard Cubic Feet per Minute - determines the weight of air to fixed or "Standard" conditions. There are several definitions of SCFM. The most common used in the United States is with the "sea-level" properties:
- 14.696 Pounds per Square Inch (psia)
- 60 Degrees Fahrenheit (oF) (520 oR)
- 0% Relative Humidity (RH)
Europeans normally use one ata and 0 oC as SCFM.
Note that the The Compressed Air & Gas Institute and PNEUROP have adopted the definition used in ISO standards with dry (0% relative humidity) air at 14.5 psia (1 bar) and 68 oF (20 oC).
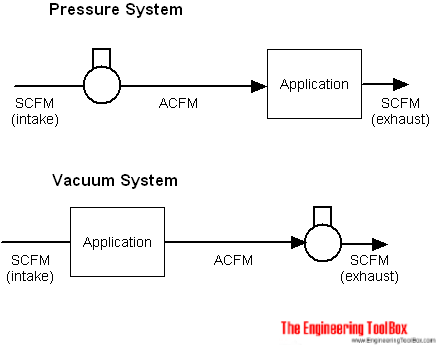
ACFM - Actual Cubic Feet per Minute
Real life "actual conditions" are seldom "standard conditions". When
- pressure is applied a volume of air - it gets smaller
- vacuum is applied to a volume of air - it expands
The actual air volume flow is often termed ACFM - Actual Cubic Feet per Minute .
The Actual Cubic Feet per Minute - ACFM - depends on the
- pressure
- temperature
- humidity
of the actual air.
The conversion from SCFM to ACFM can be expressed
ACFM = SCFM (Pstd / (Pact - Psat Φ)) (Tact / Tstd ) (1)
where
ACFM = Actual Cubic Feet per Minute
SCFM = Standard Cubic Feet per Minute
Pstd = standard absolute air pressure (psia)
Pact = absolute pressure at the actual level (psia)
Psat = saturation pressure at the actual temperature (psi)
Φ = Actual relative humidity
Tact = Actual ambient air temperature (oR)
Tstd = Standard temperature (oR)
Note!
Psat Φ ƒ < Pact
Related Mobile Apps from The Engineering ToolBox 
- free apps for offline use on mobile devices.
Online SCFM - ACFM Calculator
The calculator below can used to calculate ACFM:
Example - SCFM to ACFM
The actual CFM of a compressor operating at "non-standard" conditions like
- elevation 5000 feet (1500 m) - atmospheric pressure Pact = 12.23 psia
- temperature 80 oF - absolute temperature Tact = 540 oR
- saturation pressure Psat = 0.5069 psia
- relative humidity Φ = 80%
- demand: 100 SCFM
can be calculated as
ACFM = (100 SCFM) ((14.7 psia) / ((12.23 psia) - (0.5069 psia) (80 / 100)))((540 oR )/ (520 oR))
= 129.1
ICFM - Inlet Cubic Feet per Minute
Inlet Cubic Feet per Minute - ICFM - is used by compressor vendors to establish conditions in front of additional equipment like inlet filter, blower or booster.
When air passes through the filter there will be a pressure drop. The conversion from ACFM to ICFM can be expressed as
ICFM = ACFM (Pact / Pf) (Tf / Tact ) (2)
where
ICFM = Inlet Cubic Feet per Minute
Pf = Pressure after filter or inlet equipment (psia)
Tf = Temperature after filter or inlet equipment (oR)
Note!
The Ideal Gas Law is accurate only at relatively low pressures and high temperatures. To account for the deviation from the ideal situation, another factor is included. It is called the Gas Compressibility Factor, or Z-factor . This correction factor depends on the pressure and temperature for each gas considered.
The True Gas Law, or the Non-Ideal Gas Law, becomes:
P V = Z n R T (3)
where
Z = Gas Compressibility Factor
n = number of moles of gas present
Air Volume Conversion Factors
Air volume conversion factors related to air with temperature 70°F (21.1°C) :
| Air Temperature | Factor | |
|---|---|---|
| °F | °C | |
| 0 | -17.2 | 1.152 |
| 10 | -12.2 | 1.128 |
| 20 | -6.7 | 1.104 |
| 30 | -1.1 | 1.082 |
| 40 | 4.4 | 1.060 |
| 50 | 10.0 | 1.039 |
| 60 | 15.6 | 1.019 |
| 70 | 21.1 | 1.000 |
| 80 | 26.7 | 0.981 |
| 90 | 32.2 | 0.964 |
| 100 | 37.8 | 0.946 |
| 110 | 43.3 | 0.930 |
| 120 | 48.9 | 0.914 |
| 130 | 54.4 | 0.898 |
| 140 | 60.0 | 0.883 |
| 150 | 65.6 | 0.869 |
| 160 | 71.1 | 0.855 |
| 170 | 76.7 | 0.841 |



