Carbon Dioxide - Liquid Properties
Properties of saturated liquid Carbon Dioxide - CO2 - density, specific heat, kinematic viscosity, thermal conductivity and Prandtl number.
Properties of saturated liquid Carbon Dioxide - CO2 - density, specific heat capacity, kinematic viscosity, thermal conductivity and Prandtl number:
| Temperature - T - (oC) | Density - ρ - (kg/m3) | Specific Heat - cp - (103 J/kg K) | Thermal Conductivity - k - (W/m K) | Kinematic Viscosity - ν - (10-6 m2/s) | Prandtl's No. - Pr - |
|---|---|---|---|---|---|
| -50 | 1156 | 1.84 | 0.086 | 0.119 | 2.96 |
| -40 | 1118 | 1.88 | 0.101 | 0.118 | 2.46 |
| -30 | 1077 | 1.97 | 0.112 | 0.117 | 2.22 |
| -20 | 1032 | 2.05 | 0.115 | 0.115 | 2.12 |
| -10 | 983 | 2.18 | 0.110 | 0.113 | 2.20 |
| 0 | 927 | 2.47 | 0.105 | 0.108 | 2.38 |
| 10 | 860 | 3.14 | 0.097 | 0.101 | 2.80 |
| 20 | 773 | 5.0 | 0.087 | 0.091 | 4.10 |
| 30 | 598 | 36.4 | 0.070 | 0.080 | 28.7 |
- 1 kg/m3 = 0.0624 lb/ft3
- 1 J/(kg K) = 2.389×10-4 kcal/(kg oC) = 2.389×10-4 Btu/lbmoF
- 1 W/(m K) = 0.85984 kcal/(h m oC) = 0.5779 Btu/(ft h oF) = 0.048 Btu/(in h oF)
- 1 m2/s = 104 St = 106 cSt = 10.764 ft2/s= 38750 ft2/h
Carbon dioxide gas is produced from the combustion of coal or hydrocarbons, by fermentation of liquids and the breathing of humans and animals, and by volcanoes.
Carbon dioxide is at a low concentration in the atmosphere and acts as a greenhouse gas. Carbon dioxide is used by plants to produce carbohydrates where the photosynthesis process releases oxygen to the atmosphere.
Carbon dioxide gas is colorless and heavier than air and has a slightly irritating odor.
Freezing point is -78.5 oC where it forms carbon dioxide snow or "dry ice".
Solubility of CO2 in Water
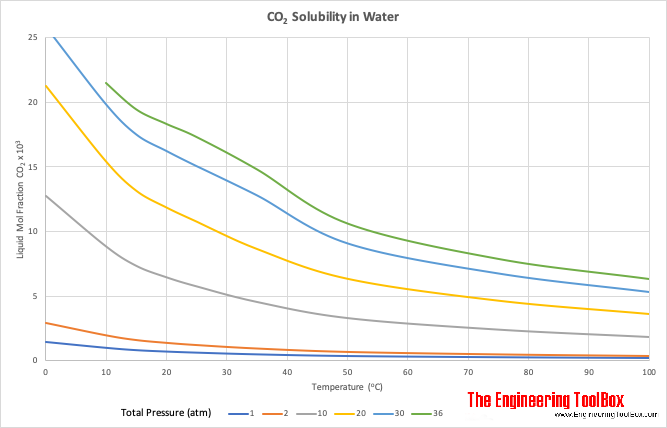
- liquid mol fraction CO2×103



