Dehumidification - Removing Moisture from Air
Dehumidifying by cooling, adsorption or absorption.
Air may be dehumidified - moisture or water removed - by
- Cooling - condensation of vapor
- Adsorption of water vapor
- Absorption of water vapor
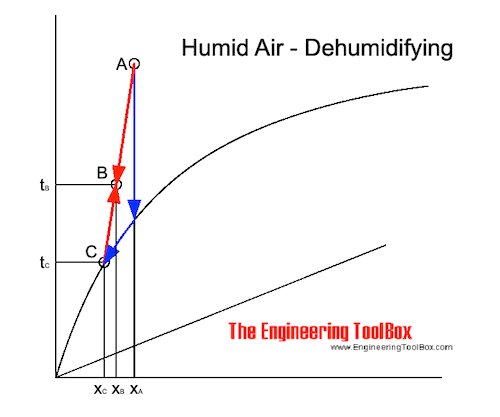
1. Cooling the air - vapor condensation
In a cooling system humidity is reduced by cooling the air below the dew point. A part of the moisture in the air is condensed and drained out.
The cooling and dehumidification process in Mollier diagram:
2. Adsorption
In an adsorption system humidity is reduced with an adsorbent material like silica gel or activated alumina.
Adsorption is a physical process in where
- moisture is condensed and held on the surface of the material
without any change of in the physical or chemical structure of the material. The adsorbent material can be reactivated by heat.
- Temperature for reactivation: 160 - 170 oC
- Heat required for reactivation: 4800 - 4800 kJ/kg water removed
Silica gel - SiO2
Silica gel - SiO2 - is a hard, adsorbent, crystalline substance and very porous. Voids are about 50 - 70% by volume and adsorbs water up to 40% of its own mass. The bulk density of silica gel is 480 - 720 kg/m3. The specific heat is 1.13 kJ/kgK.
Activated alumina
Activated alumina is about 90% aluminum oxide Al2O3 and very porous. Voids are about 50 - 70% by volume and adsorbs water up to 60% of its own mass. The bulk density is 800 - 870 kg/m3. The specific heat is 1.0 kJ/kgK.
3. Absorption
In an absorption system humidity is reduced with an absorbent material such as a calcium chloride solution.
Absorption involves a
- change in the physical or chemical structure of the material
In general - it is not easy to reactivate the material.