Critical Temperatures and Pressures for some Common Substances
Critical temperatures and pressures for some common substances like air, alcohol, ether, oxygen and more.
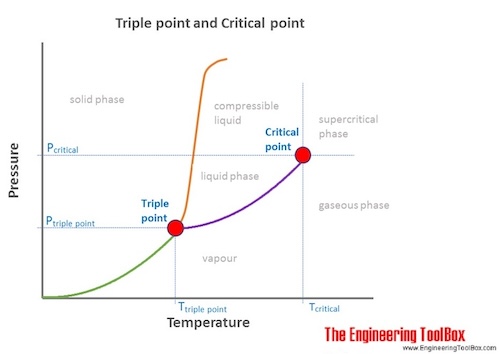
Gases can be converted to liquids by compressing the gas at a suitable temperature. However, they become more difficult to liquefy as the temperature increases because the kinetic energies of the particles that make up the gas also increase. At the critical temperature they cannot longer be liquified.
- Critical Temperature: The temperature which above, a substance can not exist as a liquid, no matter how much pressure is applied. Every substance has a critical temperature.
- Critical Pressure: The pressure required to liquify a substance vapor at its critical temperature
- Critical point: The end point of the pressure-temperature curve that designates conditions under which a liquid and its vapor can coexist. At higher temperatures, the gas cannot be liquefied by pressure alone. At the critical point, defined by the critical temperature Tc and the critical pressure pc, phase boundaries vanish.
- Triple point: The temperature and pressure at which the three phases (gas, liquid, and solid) of a substance coexist in thermodynamic equilibrium.
The table below shows critical temperature and pressure for some common substances, together with boiling temperature.
See Triple point for listing of triple point values for common substances.
| Substance | Critical temperature | Critical pressure | Boiling temperature (1 atm) | |||
|---|---|---|---|---|---|---|
| [°F] | [°C] | [psi], [lb/in2] | [bar] | [°F] | [°C] | |
| Air | -220.94 | -140.52 | 549.08 | 37.858 | - | - |
| Ammonia (NH3) | 270 | 132.4 | 1636 | 112.8 | -27.4 | -33 |
| Argon | -188 | -122 | 705.6 | 48.7 | -302.5 | -185.8 |
| Butane | 305.6 | 152 | 550.4 | 38 | 32 | 0 |
| Carbon-dioxide (CO2) | 87.8 | 31.2 | 1071.6 | 73.8 | -110 | -79 |
| Carbon-monoxide (CO) | -220.5 | -140.3 | 507.5 | 35 | -310 | -190 |
| Chlorine | 291 | 144 | 1118.7 | 77.1 | -29.3 | -34 |
| Decane | 653 | 345 | 301.7 | 20.8 | 345 | 174 |
| Dowtherm A | 465 | 32.1 | ||||
| Ethane | 90.0 | 32.2 | 708 | 48.9 | -127.4 | -88.5 |
| Ethanol (alcohol) | 467 | 242 | 914 | 63 | 173 | 78.4 |
| Ethylether | 381 | 194 | 522 | 36 | 94.2 | 34.6 |
| Ethylene | 48.9 | 9.4 | 735 | 50.7 | -272.6 | -169.2 |
| Fluorine | -200 | -129 | 808.5 | 55.8 | -307 | -188 |
| Helium | -456 | -271 | 33.2 | 2.3 | -452 | -269 |
| Hydrogen (H) | -400 | -240 | 188.2 | 13.0 | -423 | -253 |
| Hydrogen Chloride | 125 | 51.6 | 1198 | 82.7 | -121.1 | -85.1 |
| Isobutane | 274 | 135 | 529.2 | 36.5 | 10.9 | -11.7 |
| Isobutylene | 293 | 145 | 580 | 40.0 | 19.6 | -6.9 |
| Isononane | 590 | 310 | 335.1 | 23.1 | 303 | 151 |
| Methane | -117 | -82.6 | 673.3 | 46.5 | -259 | -162 |
| Nitrogen (N) | -232.6 | -147 | 492.4 | 34.0 | -321 | -195 |
| Nitrous Oxide (N2O) | 97.4 | 36.4 | 1047.6 | 72.3 | -127 | -88.5 |
| Oxygen (O2) | -181.5 | -118.6 | 732 | 50.5 | -297 | -183 |
| Phosgene | 823.2 | 56.8 | 46.9 | 8.3 | ||
| Propane | 206.1 | 96.7 | 617.4 | 42.6 | -44 | -42 |
| Propylbenzene | 689 | 365 | 464.2 | 32 | 319 | 159 |
| Propylene | 198 | 92.4 | 670.3 | 46.3 | -54 | -48 |
| Refrigerants | ||||||
| Undecane | 691 | 366 | 287.2 | 19.8 | 385 | 196 |
| Water | 705 | 374 | 3206.2 | 220.5 | 212 | 100 |
Related Topics
-
Gases and Compressed Air
Properties of air, LNG, LPG and other common gases. Pipeline capacities and sizing of relief valves. -
Thermodynamics
Work, heat and energy systems.
Related Documents
-
Acetone - Thermophysical Properties
Chemical, physical and thermal properties of acetone, also called 2-propanone, dimethyl ketone and pyroacetic acid. Phase diagram included. -
Ammonia - Dynamic and Kinematic Viscosity vs. Temperature and Pressure
Online calculator, figures and tables showing dynamic (absolute) and kinematic viscosity of gasous and liquid ammonia at temperatures ranging from -73 to 425°C (-100 to 800°F) at pressure ranging from 1 to 1000 bara (14.5 - 14500 psia) - SI and Imperial Units. -
Ammonia - Properties at Gas-Liquid Equilibrium Conditions
Figures and tables showing how the properties of liquid and gaseous ammonia changes along the boiling/condensation curve (temperature and pressure between triple point and critical point conditions). An ammonia phase diagram are included. -
Ammonia - Specific Heat vs. Temperature and Pressure
Online calculator, figures and tables showing specific heat, CP and CV, of gasous and liquid ammonia at temperatures ranging from -73 to 425°C (-100 to 800°F) at pressure ranging from 1 to 100 bara (14.5 - 1450 psia) - SI and Imperial Units. -
Ammonia - Thermal Conductivity vs. Temperature and Pressure
Online calculator, figures and tables showing thermal conductivity of liquid and gaseous ammonia at temperatures ranging -70 to 425 °C (-100 to 800 °F) at atmospheric and higher pressure - Imperial and SI Units. -
Ammonia - Thermophysical Properties
Chemical, Physical and Thermal Properties of Ammonia. Phase diagram included. -
Ammonia - Vapour Pressure at Gas-Liquid Equilibrium
Figures and table with ammonia saturation pressure at boiling points, SI and Imperial units. -
Ammonia Gas - Density vs. Temperature and Pressure
Online calculator with figures and tables showing density and specific weight of ammonia for temperatures ranging -50 to 425 °C (-50 to 800 °F) at atmospheric and higher pressure - Imperial and SI Units. -
Benzene - Thermophysical properties
Chemical, physical and thermal properties of benzene, also called benzol. Phase diagram included. -
Ethylene - Thermophysical Properties
Chemical, physical and thermal properties of ethylene, also called ethene, acetene and olefiant gas. Phase diagram included. -
Heavy Water - Thermophysical Properties
Thermodynamic properties of heavy water (D2O) like density, melting temperature, boiling temperature, latent heat of fusion, latent heat of evaporation, critical temperature and more. -
Helium - Thermophysical Properties
Chemical, Physical and Thermal Properties of Helium - He. -
Hydrogen - Thermophysical Properties
Chemical, Physical and Thermal Properties of Hydrogen - H2. -
Liquid Ammonia - Thermal Properties at Saturation Pressure
Density, specific heat, thermal conductivity, viscosity and Prandtls no. of liquid ammonia at saturation pressure. -
Liquids - Critical Pressure Ratios
Critical pressure ratios for water and other liquids. -
Non-ideal gas - Van der Waal's Equation and Constants
The van der Waals constants for more than 200 gases used to correct for non-ideal behavior of gases caused by intermolecular forces and the volume occupied by the gas particles. -
Oxygen - Thermophysical properties
Chemical, Physical and Thermal Properties of Oxygen - O2. -
The Ideal Gas Law
The relationship between volume, pressure, temperature and quantity of a gas, including definition of gas density. -
Total and Partial Pressure - Dalton's Law of Partial Pressures
How to calculate total pressure and partial pressures for gas mixtures from Ideal Gas Law. -
Triple Points Substances
Triple points for common substances. -
Water vs. Steam - Critical and Triple Points
Critical point is where vapor and liquid are indistinguishable and triple point is where ice, water and vapor coexist in thermodynamic equilibrium.




